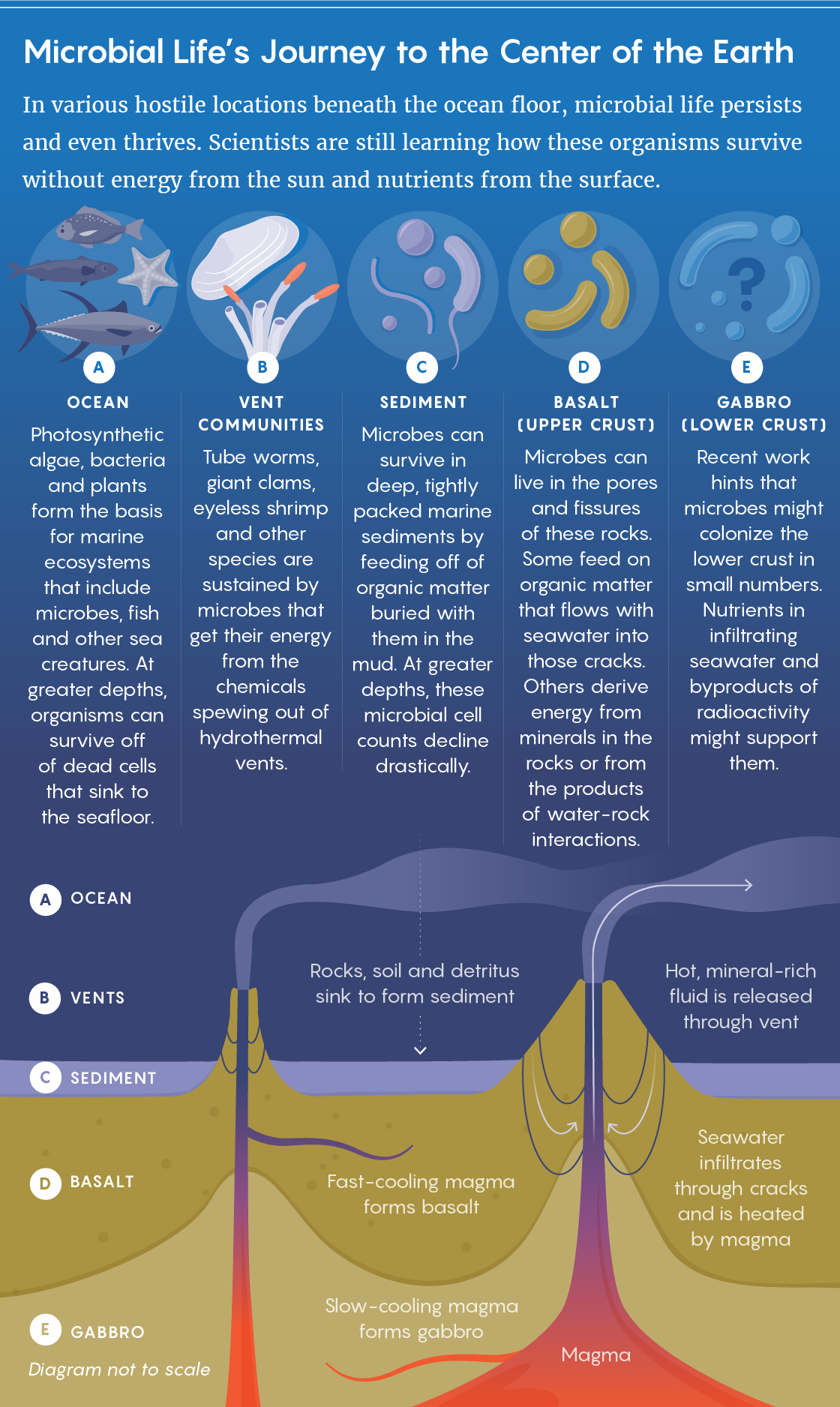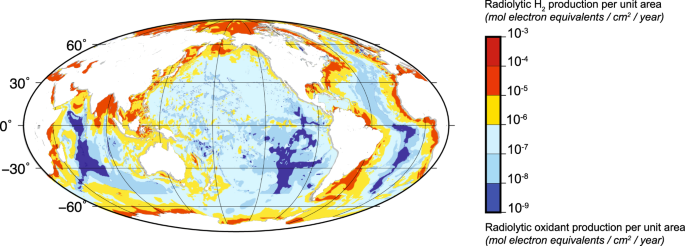
“Global distribution of production rates of radiolytic hydrogen and radiolytic oxidants in marine sediment. Sauvage┬Āet al., doi: 10.1038/s41467-021-21218-z“
SUBSURFACE BIOMES
https://doi.org/10.1016/j.gca.2020.11.026
https://doi.org/10.1038/s41467-021-21218-z
https://quantamagazine.org/radioactivity-may-fuel-life-deep-underground
Radioactivity May Fuel Life Deep Underground and Inside Other Worlds
by Jordana Cepelewicz┬Ā / ┬ĀMay 24, 2021
“Scientists poke and prod at the fringes of habitability in pursuit of lifeŌĆÖs limits. To that end, they have tunneled kilometers below EarthŌĆÖs surface, drilling outward from the bottoms of mine shafts and sinking boreholes deep into ocean sediments. To their surprise, ŌĆ£life was everywhere that we looked,ŌĆØ said┬ĀTori Hoehler, a chemist and astrobiologist at NASAŌĆÖs Ames Research Center. And it was present in staggering quantities: By various estimates, the inhabited subsurface realm has twice the volume of the oceans and holds on the order of 1030 cells, making it one of the biggest habitats on the planet, as well as one of the oldest and most diverse. Researchers are still trying to understand how most of the life down there survives. Sunlight for photosynthesis cannot reach such depths, and the meager amount of organic carbon food that does is often quickly exhausted. Unlike communities of organisms that dwell near hydrothermal vents on the seafloor or within continental regions warmed by volcanic activity, ecosystems here generally canŌĆÖt rely on the high-temperature processes that support some subsurface life independent of photosynthesis; these microbes must hang on in deep cold and darkness.
Two papers appearing in February by different research groups now seem to have solved some of this mystery for cells┬Ābeneath the continents┬Āand┬Āin deep marine sediments. They find evidence that, much as the sunŌĆÖs nuclear fusion reactions provide energy to the surface world, a different kind of nuclear process ŌĆö radioactive decay ŌĆö can sustain life deep below the surface. Radiation from unstable atoms in rocks can split water molecules into hydrogen and chemically reactive peroxides and radicals; some cells can use the hydrogen as fuel directly, while the remaining products turn minerals and other surrounding compounds into additional energy sources. Although these radiolytic reactions yield energy far more slowly than the sun and underground thermal processes, the researchers have shown that they are fast enough to be key drivers of microbial activity in a broad range of settings ŌĆö and that they are responsible for a diverse pool of organic molecules and other chemicals important to life. According to┬ĀJack Mustard, a planetary geologist at Brown University who was not involved in the new work, the radiolysis explanation has ŌĆ£opened up whole new vistasŌĆØ into what life could look like, how it might have emerged on an early Earth, and where else in the universe it might one day be found.
Barbara Sherwood Lollar set off for university in 1981, four years after the discovery of life at the hydrothermal vents. As the child of two teachers who ŌĆ£fed me on a steady diet of Jules Verne,ŌĆØ she said, ŌĆ£all of this really spoke to the kid in me.ŌĆØ Not only was studying the deep subsurface a way to ŌĆ£understand a part of the planet that had never been seen before, a kind of life that we didnŌĆÖt understand yet,ŌĆØ but it ŌĆ£clearly was going to trample [the] boundariesŌĆØ between chemistry, biology, physics and geology, allowing scientists to combine those fields in new and intriguing ways. Throughout Sherwood LollarŌĆÖs training in the 1980s and her early career as a geologist at the University of Toronto in the ŌĆÖ90s, more and more subterranean microbial communities were uncovered.
The enigma of what supported this life prompted some researchers to propose that there might be ŌĆ£a deep hydrogen-triggered biosphereŌĆØ full of cells using hydrogen gas as an energy source. (Microbes found in deep subsurface samples were often enriched with genes for enzymes that could derive energy from hydrogen.) Many geological processes could plausibly produce that hydrogen, but the best-studied ones occurred only at high temperatures and pressures. These included interactions between volcanic gases, the breakdown of particular minerals in the presence of water, and serpentinization ŌĆö the chemical alteration of certain kinds of crustal rock through reactions with water.
https://www.youtube.com/watch?v=JlcYayymVQI
By the early 2000s, Sherwood Lollar,┬ĀLi-Hung Lin┬Ā(now at National Taiwan University),┬ĀTullis Onstott of Princeton University and their colleagues were finding high concentrations of hydrogen ŌĆö ŌĆ£in some cases, stunningly high,ŌĆØ Sherwood Lollar said ŌĆö in water isolated from deep beneath the South African and Canadian crust. But serpentinization couldnŌĆÖt explain it: The kinds of minerals needed often werenŌĆÖt present. Nor did the other processes seem likely, because of the absence of recent volcanic activity and magma flows. ŌĆ£So we began to look and expand our understanding of hydrogen-producing reactions and their relationship to the chemistry and mineralogy of the rocks in these places,ŌĆØ Sherwood Lollar said.

“Bubbles of methane, hydrogen and nitrogen rise up through standing water in the Soudan Mine in Minnesota. Water radiolysis is likely to have produced at least some of these gases.”
A clue came from their discovery that the water trapped in those rocky places held not just large amounts of hydrogen but also helium ŌĆö an indicator that particles from the radioactive decay of elements like uranium and thorium were splitting water molecules. That process, water radiolysis, was first observed in Marie CurieŌĆÖs laboratory at the beginning of the 20th century, when researchers realized that solutions of radium salts generated bubbles of hydrogen and oxygen. Curie called it ŌĆ£an electrolysis without electrodes.ŌĆØ (It took a few more years for scientists to realize that the oxygen came from hydrogen peroxide created during the process.) Sherwood Lollar, Lin, Onstott and their collaborators┬Āproposed in 2006 that the microbial communities under South Africa and Canada derived the energy for their survival from hydrogen produced through radiolysis. So began their long quest to unpack how important radiolysis might be to life in natural settings. For much of the next decade, the researchers obtained samples from deep aquifers at various mining sites and related the complex chemistries of the fluids to their geological surroundings. Some of the water trapped beneath the Canadian crust had been isolated from the surface for more than 1 billion years ŌĆö perhaps even for 2 billion. Within that water were bacteria, still very much alive. ŌĆ£That had to be a completely self-sustained system,ŌĆØ Mustard observed. By the process of elimination, radiolysis looked like a possible energy source, but could there be enough of it to support life?

“A sample of ancient water found deep within Kidd Creek Mine in Ontario. In such samples, researchers have detected abiotically produced hydrogen, sulfate and organic compounds that may sustain life far below ground.”
In 2014, when Sherwood Lollar and her colleagues combined the results of nuclear chemistsŌĆÖ lab work with models of the crustŌĆÖs mineral composition, they┬Ādiscovered that radiolysis and other processes were likely to be producing a huge amount of hydrogen in the continental subsurface ŌĆö on par with the amount of hydrogen thought to arise from hydrothermal and other deep-sea environments. ŌĆ£We doubled the estimate of hydrogen production from water-rock reactions on the planet,ŌĆØ Sherwood Lollar said. Microbes could directly utilize the hydrogen produced by radiolysis, but that was only half the story: To make full use of it, they needed not just hydrogen as an electron donor, but another substance as an electron acceptor. The scientists suspected the microbes were finding that in compounds made when the hydrogen peroxide and other oxygen-containing radicals from radiolysis reacted with surrounding minerals. In┬Āwork published in 2016, they showed that radiolytic hydrogen peroxide was likely interacting with sulfides in the walls of a Canadian mine to produce sulfate, an electron acceptor. But Sherwood Lollar and her colleagues still needed proof that cells were relying on that sulfate for energy. In 2019, they finally got it. By culturing bacteria from the groundwater in mines, they were able to show that the microbes made use of┬Āboth the hydrogen and the sulfate. Water, some radioactive decay, a bit of sulfide ŌĆö ŌĆ£and then you get a sustained system of energy production that can last for billions of years ŌĆ” like an ambient pulse of habitability,ŌĆØ said┬ĀJesse Tarnas, a planetary scientist and NASA postdoctoral fellow.

“Bacteria found deep within a gold mine in South Africa that subsist on hydrogen and sulfate. Similar bacteria are believed to live at the Canadian mining sites studied by Sherwood LollarŌĆÖs group.”
In their February paper, Sherwood Lollar and her colleagues showed that radiolysis is instrumental not just in the hydrogen and sulfur cycles on Earth, but in the cycle most closely associated with life: that of carbon. Analyses of water samples from the same Canadian mine showed very high concentrations of acetate and formate, organic compounds that can support bacterial life. Moreover, measurements of isotopic signatures indicated that the compounds were being generated abiotically. The researchers hypothesized that radiolytic products were reacting with dissolved carbonate minerals from the rock to produce the large quantities of carbon-based molecules they were observing. To cement their hypothesis, Sherwood LollarŌĆÖs team needed additional evidence. It arrived just one month later. Nuclear chemists led by┬ĀLaurent Truche, a geochemist at Grenoble Alpes University in France, and┬ĀJohan Vandenborre┬Āof the University of Nantes had been independently studying radiolysis in laboratory settings. In work published in March, they pinned down┬Āthe precise mechanisms and yields┬Āof radiolysis in the presence of dissolved carbonate. They measured exact concentrations of various byproducts, including formate and acetate ŌĆö and the quantities and rates they recorded aligned with what Sherwood Lollar was seeing in the deep fractures within natural rock.
While Sherwood Lollar was conducting her field research within the continental subsurface, a handful of scientists were trying to suss out the effects of radiolysis beneath the seafloor. Chief among them was┬ĀSteve DŌĆÖHondt, a geomicrobiologist at the University of Rhode Island, who in February with his graduate student Justine Sauvage and their colleagues published the results of nearly two decadesŌĆÖ worth of detailed evidence that radiolysis is important for sustaining marine subsurface life. In 2010, DŌĆÖHondt and┬ĀFumio Inagaki, a geomicrobiologist at the Japan Agency for Marine-Earth Science and Technology, led a drilling expedition that collected samples of sub-seafloor sediments from around the globe. Subsequently, DŌĆÖHondt and Sauvage suspended dozens of sediment types in water and exposed them to different types of radiation ŌĆö and every time, they found that the amount of hydrogen produced was much greater than when pure water was irradiated. The sediments were amplifying the products of radiolysis. And ŌĆ£the yields were ridiculous,ŌĆØ DŌĆÖHondt said. In some cases, the presence of sediment in the water increased the production of hydrogen by a factor of nearly 30.
ŌĆ£Some minerals are just hotbeds of radiolytic hydrogen production,ŌĆØ DŌĆÖHondt said. ŌĆ£They very efficiently convert the energy of radiation into chemical energy that microbes can eat.ŌĆØ Yet DŌĆÖHondt and his colleagues found barely any hydrogen in the sediment cores theyŌĆÖd drilled. ŌĆ£Whatever hydrogen is being produced is disappearing,ŌĆØ DŌĆÖHondt said. The researchers think itŌĆÖs being consumed by the microbes living in the sediments. According to their models, in deep sediments more than a few million years old, radiolytic hydrogen is being produced and consumed more quickly than organic matter is ŌĆö making radiolysis of water the dominant source of energy in those older sediments. While it accounts for only 1%-2% of the total energy available in the global marine sediment environment ŌĆö the other 98% comes from organic carbon, which is mostly consumed when the sediment is young ŌĆö its effects are still quite sizable. ŌĆ£It might be slow,ŌĆØ said┬ĀDoug LaRowe, a planetary scientist at the University of Southern California, ŌĆ£but from a geologic perspective, and over geologic time ŌĆ” it starts to add up.ŌĆØ This means that radiolysis ŌĆ£is a fundamental source of bioavailable energy for a significant microbiome on earth,ŌĆØ Sauvage said ŌĆö not just on the continents but beneath the oceans, too. ŌĆ£ItŌĆÖs quite striking.ŌĆØ
The newfound scientific importance of radiolysis may not just relate to how it sustains life in extreme environments. It could also illuminate how abiotic organic synthesis may have set the stage for the origin of life ŌĆö on Earth and elsewhere. Sherwood Lollar has been invigorated by her teamŌĆÖs recent observations that, in the closed environmental system around the Canadian mines, most of the carbon-containing compounds seem to have been produced abiotically. ŌĆ£ItŌĆÖs one of the few places on the planet where the smear of life hasnŌĆÖt contaminated everything,ŌĆØ she said. ŌĆ£And those are pretty rare and precious places on our planet.ŌĆØ Part of their unique value is that they can be ŌĆ£an analogue for what might have been the prebiotic soup that our Earth might have had before life arose,ŌĆØ she continued. Even if life didnŌĆÖt arise in this kind of subsurface environment ŌĆö higher-energy regions of the planet, like hydrothermal vents, are still more probable venues for an origin story ŌĆö it provided a safe place where life could be sustained for long stretches of time, far away from the dangers found at the surface (like the meteor impacts and high levels of radiation that plagued the early Earth).
Modeling and experimental work have shown that even simple systems (consisting solely of hydrogen, carbon dioxide and sulfate, for example) can lead to extremely intricate microbial food webs; adding compounds like formate and acetate from radiolysis to the mix could significantly broaden the potential ecological landscape. And because acetate and formate can form more complex organics, they can give rise to even more diverse systems. ŌĆ£ItŌĆÖs important to see life operating with this amount of complexity,ŌĆØ said┬ĀCara Magnabosco, a geobiologist at the Swiss Federal Institute of Technology Zurich, ŌĆ£even in something that maybe you would view as very simple and very energy-poor.” ŌĆ£LetŌĆÖs say [radiolysis] can only make basic organic carbons, like formate and acetate,ŌĆØ LaRowe said. ŌĆ£If you move those compounds into a different environmental setting, perhaps they can react there to form something else. They become starter or feeder material for more complex reactions in a different setting.ŌĆØ That might even help bring scientists closer to understanding how amino acids and other important building blocks of life arose.
Sherwood Lollar is now collaborating with other scientists, including colleagues at the┬ĀCIFAR Earth 4D project, to study how the organic molecules present in the ancient Canadian water might ŌĆ£complexifyŌĆØ the chemistry at hand. In work theyŌĆÖre hoping to publish later this year, ŌĆ£we show how the coevolution of organics and minerals is key for the diversification of these organic compounds,ŌĆØ said┬ĀB├®n├®dicte Menez, a geobiologist at the Paris Institute of Earth Physics and one of the leaders of the research. Her aim is to determine how more complicated organic structures could form and subsequently play a role in some of the earliest microbial metabolisms. Astrobiologists are also realizing how crucial it might be to consider radiolysis when constraining the habitability of planets and moons throughout the solar system and the rest of the galaxy. Sunlight, high temperatures and other conditions might not be strictly needed to sustain extraterrestrial life. Radiolysis should be practically ubiquitous on any rocky planet that has water in its subsurface.
Take Mars. In a pair of studies,┬Āone published a couple of years ago┬Āand┬Āthe other last month, Tarnas, Mustard, Sherwood Lollar and other researchers translated quantitative work being done on radiolysis on Earth to the Martian subsurface. They found that based on the planetŌĆÖs mineral composition and other parameters, Mars today might be able to sustain microbial ecosystems akin to those on Earth ŌĆö with radiolysis alone. The scientists identified regions of the planet where the microbial concentration would likely be greatest, which could guide where future missions should be targeted. ŌĆ£ItŌĆÖs really fascinating to me,ŌĆØ Inagaki said, ŌĆ£as we are now in an era where particle physics is necessary to study microbial life in EarthŌĆÖs planetary interior and other worlds in the universe.ŌĆØ

“As scientists continue to find microbes deeper and deeper beneath the ocean floor, they are beginning to suspect that the right combination of rocks and water might be enough to sustain life almost anywhere.”
WATER RADIOLYSIS
https://nature.com/articles/nature14017
https://mdpi.com/2073-4441/3/1/235/htm
https://quantamagazine.org/inside-undersea-rocks-life-thrives-without-sun
Inside Deep Undersea Rocks, Life Thrives Without the Sun
by Jordana Cepelewicz┬Ā /┬Ā May 13, 2020
“Microbial life, almost unbelievably resilient, abides in boiling hot springs and bone-dry deserts, in pools of acid and polar ice, kilometers up into the sky and kilometers below the ocean floor. And while scientists are eager to uncover microbes in even less familiar territories beyond our solar system, itŌĆÖs the last Earth-bound frontier on that list ŌĆö the deep subsurface ŌĆö where theyŌĆÖre now making exciting progress in their efforts to probe lifeŌĆÖs extreme adaptability. Lightless, barren of essential nutrients and crushed under inconceivable pressures, the deep subsurface seems unrelentingly inhospitable, yet it is shaping up to be one of EarthŌĆÖs biggest habitats. Moreover, its strangeness is forcing scientists to reckon with biological systems that operate on completely different energy sources and time scales from those that we surface dwellers are accustomed to.
Scientists have spent decades studying how and where microbes persist and even thrive beneath the oceans, far removed from the sun. Most of that work has focused on marine sediments, the tightly packed mud and detritus that in places extends for kilometers beneath the water. But thereŌĆÖs also the volcanic rock below that, the crust itself. The life in those rocks is much more difficult to access and analyze, and samples are scarce. ŌĆ£We do not have a map of the deep subsurface microbial landscape right now,ŌĆØ said┬ĀKaren Lloyd, a microbiologist at the University of Tennessee, Knoxville. As a result, she noted, researchers studying its diverse range of environments canŌĆÖt even make simple generalizations equivalent to ŌĆ£forests have trees and fish swim in the oceans.ŌĆØ But a handful of new discoveries has finally opened a window onto that landscape and the microbes that inhabit it. It has also offered a glimpse into the very origin and evolution of life, both on this planet and possibly elsewhere in the universe.
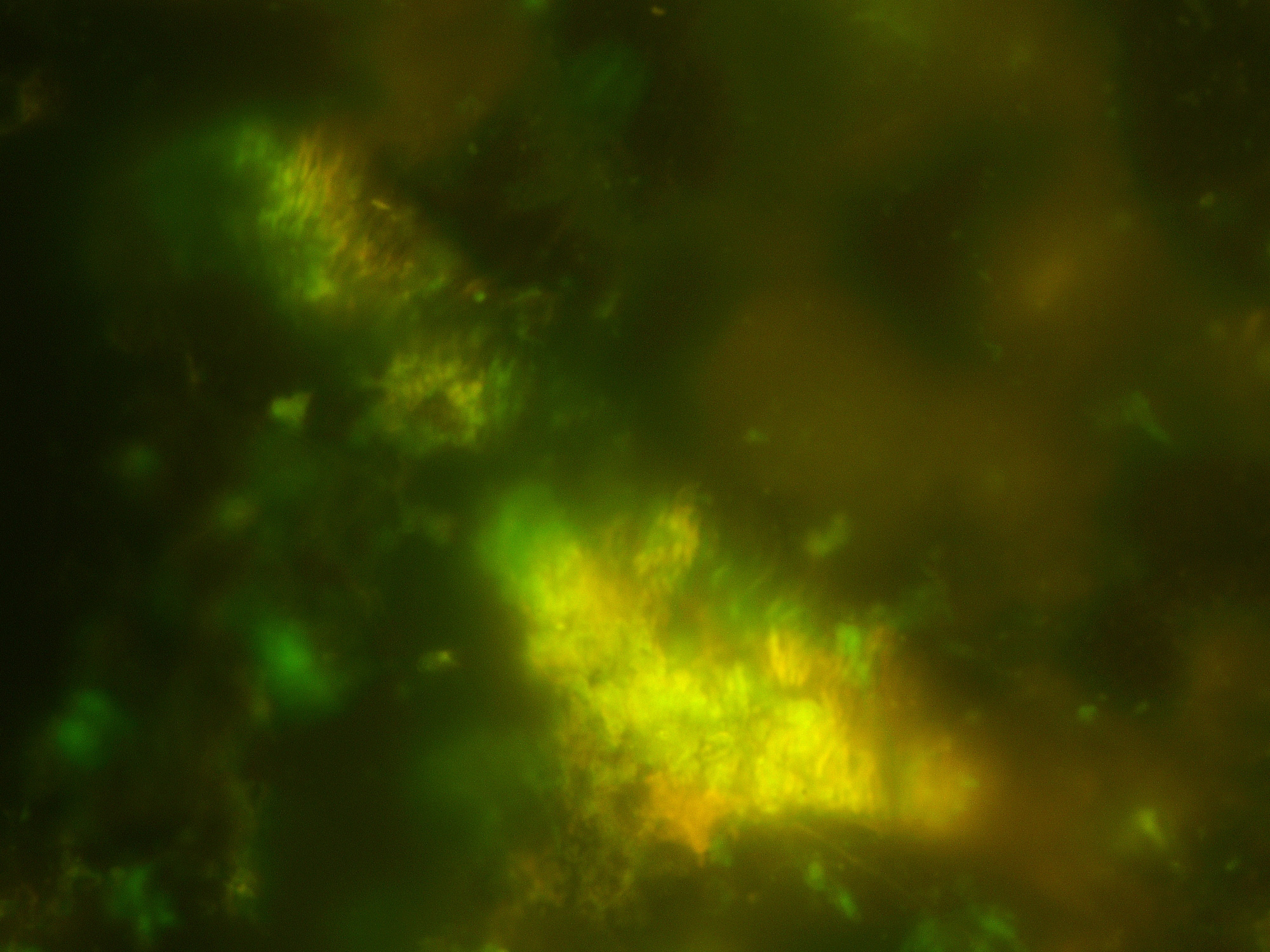
“Fluorescence microscopy shows the remarkable density of bacteria growing inside basaltic rocks sampled from below the seafloor. The cells are stained green; yellow shows where the cells are inside aggregates of clay minerals.”
EarthŌĆÖs crust has largely been the purview of geologists. The first indication that it might also be of interest to microbiologists came in 1926, when┬Āresearchers reported the presence of bacteria┬Āin the deep oil wells of Illinois. Such findings, however, wouldnŌĆÖt be taken seriously for decades: Contamination in the samples seemed far more likely than the possibility that anything could live so disconnected from the sun-driven photosynthesis that supports life everywhere else on the planet. (It didnŌĆÖt help that in the 1950s, a pair of microbiologists┬Āperformed a series of experiments that led them to pin the bottom of the biosphere to sediments just a few meters below the seafloor.) In 1977, that all began to change. Scientists boarded a submarine to explore a spreading ocean ridge between two tectonic plates near the Gal├Īpagos Islands, and discovered the first hydrothermal vents. There, the Earth belched out billows of black smoke, as scalding-hot, mineral-rich fluids spewed from cracks in the rock and mixed with the extremely cold seawater. Around those vents, an entire ecosystem flourished, including tube worms, giant clams and eyeless shrimp ŌĆö and, it turned out, lots and lots of microbes that sustained them. ŌĆ£There, in these dark, deep parts of the ocean where they didnŌĆÖt expect to find life, certainly they did,ŌĆØ said┬ĀBarbara Sherwood Lollar, a geologist at the University of Toronto. And it was ŌĆ£literally life as we didnŌĆÖt know it.ŌĆØ

“Barbara Sherwood Lollar looks for signs of life within the ŌĆ£hidden hydro-geosphereŌĆØ ŌĆö water deep underground that may have been isolated from the surface for millions or even billions of years.”
For the first time, scientists realized there could be ecosystems on Earth that did not depend on the sun. The microbes were powered not by solar energy, but by minerals and chemicals released at the vents. They challenged what life could be, and where its limits lay. In the 1980s and ŌĆÖ90s, scientists began to uncover further evidence of an unlit but populated kingdom: Rocks beneath both the continents and oceans exhibited weathering that seemed unlikely to be a result of abiotic reactions alone. Drilling projects on land and at sea revealed living cells and DNA sequences in all sorts of environments, leading researchers to speculate that subsurface microbes constituted a hidden, pervasive majority, greatly outnumbering the microbial cells found in the world above. Current estimates, in fact, put┬Āthe number of subsurface microbes┬Āon the order of 1030┬Ācells, an order of magnitude higher than the number of microbes thought to dwell in soil or the open ocean. ŌĆ£The deep biosphere,ŌĆØ said┬ĀVirginia Edgcomb, a marine microbiologist at the Woods Hole Oceanographic Institution in Massachusetts, ŌĆ£is extensive. What captivated the attention of microbiologists was that [potential] extent.ŌĆØ And everywhere theyŌĆÖve looked in that extensive realm since ŌĆö no matter how deep or seemingly nutrient-poor ŌĆötheyŌĆÖve found life.
The domain below the seafloor can be divided into two distinct regimes: sediment and rock. The former comprises the mud and detritus that accumulates at the bottom of the ocean. This layer resembles a dense sponge in structure: Although 90 percent of its weight might be water, nothing can flow through it efficiently; fluids and chemical compounds slowly diffuse through it instead. Microbial cells essentially get buried there, along with whatever matter they might use for energy. In shallow areas, particularly near coasts where nutrients are more abundant, this buried life thrives: Millions or even a billion microbial cells may dwell in a cubic centimeter of that sediment. As researchers dig deeper, they find fewer cells. And yet they always seem to find something. TheyŌĆÖve dug into sediments┬Āas far as 2,500 meters below the seafloor ŌĆö where theyŌĆÖve uncovered just a few cells per cubic centimeter, approaching the very limits of their detection ability. These cells barely seem alive, at least by our standards. They live very, very slowly, rarely dividing, their energy consumption at times six orders of magnitude lower than that of cells living in surface habitats. ŌĆ£It might take them 100 years or 1,000 years to divide just once,ŌĆØ said┬ĀMartin Fisk, an ocean ecologist at Oregon State University. ŌĆ£TheyŌĆÖre very slowly keeping themselves going.ŌĆØ

“A piece of the basaltic rock retrieved from a core sample, prior to processing and analysis for any cells it might contain.”
Steve DŌĆÖHondt, an oceanographer at the University of Rhode Island, calls them ŌĆ£zombie cells.ŌĆØ But they are, indeed, alive ŌĆö theyŌĆÖre simply not operating at familiar time scales. Our observations of the cellsŌĆÖ activity might be akin to a mayflyŌĆÖs experience of the life of a tree; the insect dies well before it can gain any true understanding of how the tree develops and sustains itself. But something different might be happening in the basalt rocks that lie beneath the sediment. Unlike the hard-packed mud, those rocks have pores and cracks and fissures through which seawater circulates ŌĆö and with it, organic matter that microbes can feed on. Unfortunately, itŌĆÖs also much more challenging to penetrate and obtain uncontaminated samples from that rock. While sediment can be sampled by essentially driving a pipe vertically into the mud, the rock is too hard for that. Scientists instead have to use a drill, as well as a fluid mixture of seawater and mud to lubricate it ŌĆö and that process contaminates the edges and crevices of the samples with biological material from higher up. So to investigate what might be living inside the rocks, microbiologists have to clean them, rinse them in alcohol, flame their surface and break them open. Even then, these methods tend to uncover very limited numbers of cells, and without accompanying photographic evidence, ŌĆ£itŌĆÖs always very hard to prove that what is being sampled is really from there, and not contamination,ŌĆØ said┬ĀWilliam Orsi, a geomicrobiologist at the University of Munich.

“A prepared rock sample from a deep-sea drilling expedition. The geoscientist Yohey Suzuki and his colleagues developed a novel way to identify and count the cells living inside these rocks.”
Most of the handful of studies that have examined life in basalts have focused on rocks near hydrothermal vents or newly made seafloor ŌĆö relatively young rocks that are only up to 8 million years old. ThatŌĆÖs because itŌĆÖs in younger rocks that seawater can most easily circulate and replenish the microbesŌĆÖ food supply. Those rocks are also more chemically reactive, and certain ŌĆ£rock-eatingŌĆØ microbes can use those reactions as an energy source, too. But as basalt cools and moves away from where it was formed, further along the ocean floor toward subduction zones, its many cracks and pores fill up with precipitated minerals, preventing this more active fluid flow and leading to a more isolated system. It was unclear whether life could survive under such conditions ŌĆö until now. Last month, a team of scientists in Japan┬Āreported life in basalts collected by a 2010 drilling expedition that had reached 120 meters into the crust beneath the ocean, to rocks that were between 33 million and 104 million years old. The age of the rocks alone was exciting, according to Lloyd, who did not participate in the study. ŌĆ£When it formed, dinosaurs were walking the Earth.ŌĆØ

“Yohey Suzuki and his colleagues recently found evidence of high concentrations of microbes living inside sub-seafloor basalts. Some of those rock formations were over 100 million years old.”
But even more tantalizing were the densities at which the microbes were growing in the rocks. The researchers, using a new technique to precisely enumerate the cells in their samples, found that the microbes were concentrated in a particular subset of the basaltŌĆÖs mineral-filled fissures. There, they formed something like a biofilm, reaching counts of 10 billion cells per cubic centimeter. ŌĆ£[We] didnŌĆÖt think that there [would be] so many cells in old, cold, hard rocks,ŌĆØ said┬ĀYohey Suzuki, a geoscientist at the University of Tokyo and the lead author of the study. The researchers were also able to map which types of minerals the cells did and did not associate with. They posit that the bacteria survived by living off of organic matter trapped in those minerals. ŌĆ£That opens up much, much more of the seafloor that we now know should host microbial life,ŌĆØ said┬ĀJason Sylvan, a biogeochemist at Texas A&M University who was not involved with the research. In fact, it suggests that the recipe for life might be less stringent than previously expected. Perhaps all thatŌĆÖs needed to sustain it is a combination of rock and fluid flow.

“Water pouring into this mine shaft attests to the abundant liquid circulating through fissures and fractures far below the EarthŌĆÖs surface. Scientists can analyze how long that water has been isolated from the surface world.”
ThatŌĆÖs not an entirely new idea. In places like South Africa and Canada, work in terrestrial deep mines ŌĆö which descend into rocks that are billions of years older than the oldest seafloor basalts ŌĆö has hinted at this for the past decade. Sherwood Lollar, along with Princeton UniversityŌĆÖs┬ĀTullis Onstott┬Āand other colleagues, have ventured into those mines to study what they call ŌĆ£the hidden hydro-geosphere,ŌĆØ systems of water isolated deep underground on long geological time scales. In some cases, theyŌĆÖve found water that hasnŌĆÖt been exposed to surface environmental factors in millions or┬Āeven billions of years. And in that billion-year-old water, the researchers┬Āhave found life. TheyŌĆÖve also found evidence that those microbes persist by getting energy from an abiotic process called radiolysis, during which radiation released by the rocks reacts with water in the system to release hydrogen, which the cells can then use in various forms as fuel. ThatŌĆÖs posed an intriguing question for scientists: Could radiolysis be an alternative process driving much of subsurface life? Given that radiolysis occurs everywhere, ŌĆ£it could also be supporting the ocean deep life,ŌĆØ Orsi said. ŌĆ£No one knows.ŌĆØ
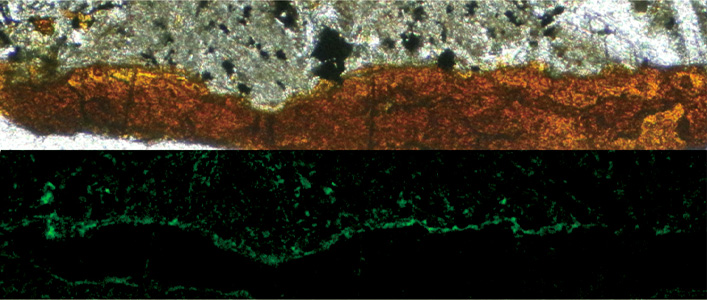
“Two views of a cross-section through old sub-surface basalt. At top, in light microscopy, the basalt appears gray and the clay minerals filling a crevice are orange. At bottom, under fluorescence, stained bacteria on the surface of the minerals glow green. Suzuki et al. 2020, DOI: 10.1038/s42003-020-0860-1“
ItŌĆÖs much more difficult to obtain evidence of that in marine environments, where thereŌĆÖs more fluid flow and less obvious isolation. But DŌĆÖHondt, who participated in the study of the old sub-seafloor basalts, posits that the bacteria recently uncovered there might rely on radiolysis, too. ŌĆ£ItŌĆÖs hard to imagine enough organic matter bleeding in there to support these cell densities,ŌĆØ he said. Perhaps radiolysis is part of the picture as well. The relative isolation of the habitat, thanks to the mineral filling, makes it an intriguing subject for future study. At the very least, ŌĆ£we basically learned from the [terrestrial] work that life can fit into almost any crevice that has water,ŌĆØ said┬ĀJennifer Biddle, a marine scientist at the University of Delaware. Now, the kind of work done by Suzuki, DŌĆÖHondt and their team is showing ŌĆ£that the marine environment is following the same rules as the terrestrial environment: As long as you have space and water, youŌĆÖve got a chance of something going,ŌĆØ she said. Maybe that applies to radiolysis, too ŌĆö maybe itŌĆÖs to subsurface life what photosynthesis is to surface life.
Regardless, it teaches scientists an important lesson: that to understand any deep-sea life, they canŌĆÖt just look at the rocks. They also have to study the water ŌĆö how it flows, or doesnŌĆÖt. Several groups are now doing this in basalt systems throughout the world. And it offers a new way to visualize potential limits to life. Traditionally, researchers hypothesize that life is most frequently limited by temperature. Above around 120 degrees Celsius, even the hardiest microbial cells break apart and die. But another major factor seems to be this all-important fluid flow. As long as the temperature stays cool enough, life ŌĆ£may go as deep as seawater can penetrate,ŌĆØ DŌĆÖHondt said. Perhaps most important, such studies of fluid flow offer a new lens through which to scrutinize lifeŌĆÖs origins and evolution.
Since the discovery of hydrothermal vents, Sherwood Lollar said, ŌĆ£weŌĆÖve seen a shift from the idea of DarwinŌĆÖs ŌĆśwarm little pondŌĆÖŌĆØ as far as the origin of life is concerned. Now, we may need to consider not a warm little pond or a hydrothermal vent, but what Onstott has called ŌĆ£a warm little fractureŌĆØ ŌĆö particularly when one takes into account the bombardments and other hostile environmental factors that would have characterized the surface of early Earth. ŌĆ£You have volcanic rock and water, and you can get life going,ŌĆØ said Fisk, who recently┬Āmodeled this possibility. That is, itŌĆÖs possible that life might have gotten its start on the surface of the Earth, where it found creative ways to survive and spread, including to deeper environments. But itŌĆÖs also possible that life began underground, at some fortuitous juncture between rock and water ŌĆö eventually also making its way to the surface and figuring out how to harness the sunŌĆÖs energy. (On that note, photosynthesis-dependent surface life and radiolysis-dependent subsurface life have so far been found to have an ancient, shared ancestry ŌĆö but some researchers are intrigued by the possibility that life could have evolved more than once on Earth, in a ŌĆ£second genesis.ŌĆØ)
That, in turn, has significant implications for the search for life on Mars, SaturnŌĆÖs moon Enceladus and exoplanets beyond our solar system. Given the prevalence of water and volcanic rock throughout the universe, ŌĆ£life could have started anywhere,ŌĆØ Fisk said. DŌĆÖHondt agreed. ŌĆ£From a literally universal perspective, it opens up the potential for sustaining life on all kinds of planets,ŌĆØ he said. ŌĆ£There could be life on other worlds thatŌĆÖs independent of photosynthesis,ŌĆØ thriving beneath the surface, out of sight. Even more intriguing, perhaps life can evolve and adapt down there, too ŌĆö perhaps very differently from how it has on the surface. Given how slowly the cells beneath the ocean floor grow, could they also be adapting on much more gradual time scales?
A couple of preliminary studies, conducted in younger sediments, have not found evidence for such evolution ŌĆö but those only dealt with one type of deep subsurface environment, and even then, they went back only around 10,000 years. That might not be enough time for a slower kind of natural selection to have played a role. ŌĆ£What if you go back a million years or 10 million years?ŌĆØ Orsi asked. Older sediments ŌĆö as well as other sub-seafloor environments, such as the basalts examined by Suzuki and DŌĆÖHondt ŌĆö might hold fresh insights. ŌĆ£When you expand the possibilities to greater and greater time scalesŌĆØ and contexts, Lloyd said, ŌĆ£then you have a wider array of possible drivers for evolution.ŌĆØ
Even now, scientists are continuing to push further, deeper. A┬Āpaper published in┬ĀNature in March, spearheaded by Edgcomb, detailed the results from one of the most ambitious such pushes to date. They drilled nearly 800 meters below the seafloor at a location where the lower ocean crust bulges up closer to the surface. There, they sampled gabbro rocks, which are formed when magma cools more slowly: TheyŌĆÖre typically found beneath basalt and considered a sort of window into the mantle, as well as into what an earlier EarthŌĆÖs rock environment might have looked like. And in those rocks, the scientists reported trace numbers of cells ŌĆö which once again seemed to be surviving off nutrients they gleaned from the flow of seawater. Although other researchers, including Orsi, have raised concerns about the possibility that those samples were contaminated, they also expect future analyses to uncover life down there. No matter what, Edgcomb said, echoing the famous line from┬ĀJurassic Park, ŌĆ£life finds a way.ŌĆØ

“Scientists transmit electric current into the water to lure microbes.”
METAL-EATING MICROBES
https://pubmed.ncbi.nlm.nih.gov/25900658
https://quantamagazine.org/electron-eating-microbes
https://quantamagazine.org/how-to-grow-metal-eating-microbes
How to Grow Metal-Eating Microbes
by Jeanette Kazmierczak┬Ā /┬Ā June 21, 2016
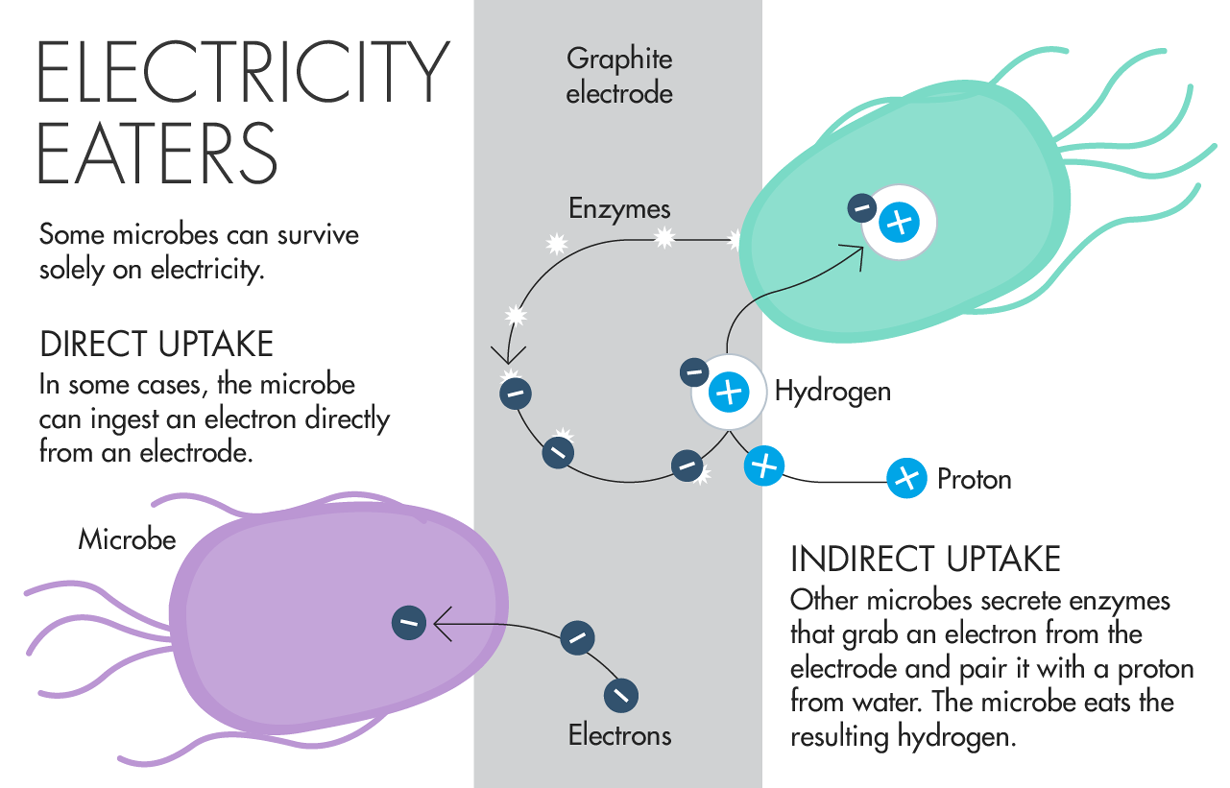
“At first glance, the sunny, blue waters off Southern CaliforniaŌĆÖs Catalina Island donŌĆÖt seem to have much in common with an old gold mine in South DakotaŌĆÖs Black Hills. YouŌĆÖd have to get down to the microbial level before you spotted the similarities. Both the waters of the gold mine and the seafloor sediment off Catalina are mineral-rich, oxygen-poor environments home to a strange class of microbe ŌĆö the kind that eat metals to get electrons. All life on Earth needs a source of electrons to make and store energy; the trick is getting them across the fatty, insulating cellular membrane. Humans eat food, smuggling electrons across in the form of glucose, then breathing them back out attached to carbon dioxide. But metal-eating microbes apparently get their electrons directly from the inhospitable, mineral-rich surfaces they call home.
Studying how, exactly, the microbes do this has been difficult because theyŌĆÖre hard to grow in the lab. But in 2015, Stanford University microbiologist┬ĀAlfred Spormann┬Āand his team┬Āshowed that one kind of metal-eating microbe can survive on the surface of an electrode by secreting an enzyme. The enzyme partners an electron from the electrode with a proton from water to make a hydrogen atom, which the microbe can absorb. But could some kinds of microbes take up electrons directly, without having to partner it with anything? Many scientists believe so, and Spormann claims to have found one already, although the results havenŌĆÖt been published. ŌĆ£Using electrodes as proxies for minerals has helped us open and expand this field,ŌĆØ said┬ĀAnnette Rowe, a postdoctoral researcher at the University of Southern California. ŌĆ£Now we have a way to grow the bacteria and monitor their respiration and really have a look at their physiology.ŌĆØ Life-forms like these could also teach us about the possibilities for life on other planets. The South Dakota gold mine project, led by┬ĀMoh El-Naggar at the University of Southern California, is funded by NASAŌĆÖs Astrobiology Initiative. He said that Mars, iron-rich with flowing water beneath the surface, has all the ingredients electron-eating life would want.”

“Scientists have figured out how microbes can suck energy from rocks. Such life-forms might be more widespread than anyone anticipated.”
ROCK-EATING MICROBES
https://iopscience.iop.org/article/10.1088/1361-6528/ab6ab5
https://newscientist.com/electric-life-forms-live-on-pure-energy
https://quantamagazine.org/electron-eating-microbes-found-in-odd-places
New Life Found That Lives Off Electricity
by Emily Singer┬Ā /┬Ā June 21, 2016
“Last year, biophysicist Moh El-Naggar┬Āand his graduate student┬ĀYamini Jangir┬Āplunged beneath South DakotaŌĆÖs Black Hills into an old gold mine that is now more famous as a home to a┬Ādark matter detector. Unlike most scientists who make pilgrimages to the Black Hills these days, El-Naggar and Jangir werenŌĆÖt there to hunt for subatomic particles. They came in search of life. In the darkness found a mile underground, the pair traversed the mineŌĆÖs network of passages in search of a rusty metal pipe. They siphoned some of the pipeŌĆÖs ancient water, directed it into a vessel, and inserted a variety of electrodes. They hoped the current would lure their prey, a little-studied microbe that can live off pure electricity.
The electricity-eating microbes that the researchers were hunting for belong to a larger class of organisms that scientists are only beginning to understand. They inhabit largely uncharted worlds: the bubbling cauldrons of deep sea vents; mineral-rich veins deep beneath the planetŌĆÖs surface; ocean sediments just a few inches below the deep seafloor. The microbes represent a segment of life that has been largely ignored, in part because their strange habitats make them incredibly difficult to grow in the lab.
Yet early surveys suggest a potential microbial bounty. A recent sampling of microbes collected from the seafloor near Catalina Island, off the coast of Southern California, uncovered a surprising variety of microbes that consume or shed electrons by eating or breathing minerals or metals. El-NaggarŌĆÖs team is still analyzing their gold mine data, but he says that their initial results echo the Catalina findings. Thus far, whenever scientists search for these electron eaters in the right locations ŌĆö places that have lots of minerals but not a lot of oxygen ŌĆö they find them.
As the tally of electron eaters grows, scientists are beginning to figure out just how they work. How does a microbe consume electrons out of a piece of metal, or deposit them back into the environment when it is finished with them? A study published last year revealed the way that one of these microbes catches and consumes its electrical prey. And not-yet-published work suggests that some metal eaters transport electrons directly across their membranes ŌĆö a feat once thought impossible. Though eating electricity seems bizarre, the flow of current is central to life. All organisms require a source of electrons to make and store energy. They must also be able to shed electrons once their job is done. In describing this bare-bones view of life, Nobel Prize-winning physiologist Albert Szent-Gy├Črgyi once said, ŌĆ£Life is nothing but an electron looking for a place to rest.ŌĆØ
Humans and many other organisms get electrons from food and expel them with our breath. The microbes that El-Naggar and others are trying to grow belong to a group called lithoautotrophs, or rock eaters, which harvest energy from inorganic substances such as iron, sulfur or manganese. Under the right conditions, they can survive solely on electricity. The microbesŌĆÖ apparent ability to ingest electrons ŌĆö known as direct electron transfer ŌĆö is particularly intriguing because it seems to defy the basic rules of biophysics. The fatty membranes that enclose cells act as an insulator, creating an electrically neutral zone once thought impossible for an electron to cross. ŌĆ£No one wanted to believe that a bacterium would take an electron from inside of the cell and move it to the outside,ŌĆØ said┬ĀKenneth Nealson, a geobiologist at the University of Southern California, in a┬Ālecture┬Āto the Society for Applied Microbiology in London last year.
https://www.youtube.com/watch?v=qBOWuMz-RaU
In the 1980s, Nealson and others discovered a surprising group of bacteria that can expel electrons directly onto solid minerals. It took until 2006 to discover the molecular mechanism behind this feat: A┬Ātrio of specialized proteins sits in the cell membrane, forming a conductive bridge that transfers electrons to the outside of cell. (Scientists still debate whether the electrons traverse the entire distance of the membrane unescorted.) Inspired by the electron-donators, scientists began to wonder whether microbes could also do the reverse and directly ingest electrons as a source of energy.
Researchers focused their search on a group of microbes called methanogens, which are known for making methane. Most methanogens arenŌĆÖt strict metal eaters. But in 2009,┬ĀBruce Logan, an environmental engineer at Pennsylvania State University, and collaborators showed for the first time that a methanogen could survive using only┬Āenergy from an electrode. The researchers proposed that the microbes were directly sucking up electrons, perhaps via a molecular bridge similar to the ones the electron-producers use to shuttle electrons across the cell wall. But they lacked direct proof. Then last year,┬ĀAlfred Spormann, a microbiologist at Stanford University, and collaborators poked a hole in LoganŌĆÖs theory. They┬Āuncovered a way┬Āthat these organisms can survive on electrodes without eating naked electrons.
The microbe Spormann studied,┬ĀMethanococcus maripaludis, excretes an enzyme that sits on the electrodeŌĆÖs surface. The enzyme pairs an electron from the electrode with a proton from water to create a hydrogen atom, which is a well-established food source among methanogens. ŌĆ£Rather than having a conductive pathway, they use an enzyme,ŌĆØ said┬ĀDaniel Bond, a microbiologist at the University of Minnesota Twin Cities. ŌĆ£They donŌĆÖt need to build a bridge out of conductive materials.ŌĆØ Though the microbes arenŌĆÖt eating naked electrons, the results are surprising in their own right. Most enzymes work best inside the cell and rapidly degrade outside.

“Strategy to isolate electroactive microorganisms from the environment”
ŌĆ£WhatŌĆÖs unique is how stable the enzymes are when they [gather on] the surface of the electrode,ŌĆØ Spormann said. Past experiments suggest these enzymes are active outside the cell for only a few hours, ŌĆ£but we showed they are active for six weeks.ŌĆØ Spormann and others still believe that methanogens and other microbes can directly suck up electricity, however. ŌĆ£This is an alternative mechanism to direct electron transfer, it doesnŌĆÖt mean direct electron transfer canŌĆÖt exist,ŌĆØ said┬ĀLargus Angenent, an environmental engineer at Cornell University, and president of the┬ĀInternational Society for Microbial Electrochemistry and Technology. Spormann said his team has already found a microbe capable of taking in naked electrons. But they havenŌĆÖt yet published the details.
Only a tiny fraction ŌĆö perhaps 2 percent ŌĆö of all the planetŌĆÖs microorganisms can be grown in the lab. Scientists hope that these new approaches ŌĆö growing microbes on electrodes rather than in traditional culture systems ŌĆö will provide a way to study many of the microbes that have been so far impossible to cultivate. ŌĆ£Using electrodes as proxies for minerals has helped us open and expand this field,ŌĆØ said┬ĀAnnette Rowe, a postdoctoral researcher at USC working with El-Naggar. ŌĆ£Now we have a way to grow the bacteria and monitor their respiration and really have a look at their physiology.ŌĆØ Rowe has already had some success. In 2013, she went on a microbe prospecting trip to the iron-rich sediments that surround CaliforniaŌĆÖs Catalina Island.

“Sediment free bioreactor schematic including three electrode system“
She identified at least┬Ā30 new varieties of electric microbes in a study published last year. ŌĆ£They are from very diverse groups of microbes that are quite common in marine systems,ŌĆØ Rowe said. Before her experiment, no one knew these microbes could take up electrons from an inorganic substrate, she said. ŌĆ£ThatŌĆÖs something we werenŌĆÖt expecting.ŌĆØ Just as fishermen use different lures to attract different fish, Rowe set the electrodes to different voltages to draw out a rich diversity of microbes. She knew when she had a catch because the current changed ŌĆö metal eaters generate a negative current, as the microbes suck electrons from the negative electrode.

“Yamini Jangir, then a graduate student in Moh El-NaggarŌĆÖs lab at the University of Southern California, collects water from a pipe at the Sanford Underground Research Facility nearly a mile underground.”
The different varieties of bacteria that Rowe collected thrive under different electrical conditions, suggesting they employ different strategies for eating electrons. ŌĆ£Each bacteria had a different energy level where electron uptake would happen,ŌĆØ Rowe said. ŌĆ£We think that is indicative of different pathways.ŌĆØ Rowe is now searching new environments for additional microbes, focusing on fluids from a deep spring with low acidity. SheŌĆÖs also helping with El-NaggarŌĆÖs gold mine expedition. ŌĆ£We are trying to understand how life works under these conditions,ŌĆØ said El-Naggar. ŌĆ£We now know that life goes far deeper than we thought, and thereŌĆÖs a lot more than we thought, but we donŌĆÖt have a good idea for how they are surviving.ŌĆØ
El-Naggar emphasizes that the field is still in its infancy, likening the current state to the early days of neuroscience, when researchers poked at frogs with electrodes to make their muscles twitch. ŌĆ£It took a long time for the basic mechanistic stuff to come out,ŌĆØ he said. ŌĆ£ItŌĆÖs only been 30 years since we discovered that microbes can interact with solid surfaces.ŌĆØ Given the bounty from these early experiments, it seems that scientists have only scratched the surface of the microbial diversity that thrives beneath the planetŌĆÖs shallow exterior. The results could give clues to the┬Āorigins of life on Earth┬Āand beyond. One theory for the emergence of life suggests it originated on mineral surfaces, which could have concentrated biological molecules and catalyzed reactions. New research could fill in one of the theoryŌĆÖs gaps ŌĆö a mechanism for transporting electrons from mineral surfaces into cells.
Moreover, subsurface metal eaters may provide a blueprint for life on other worlds, where alien microbes might be hidden beneath the planetŌĆÖs shallow exterior. ŌĆ£For me, one of the most exciting possibilities is finding life-forms that might survive in extreme environments like Mars,ŌĆØ said El-Naggar, whose gold mine experiment is funded by NASAŌĆÖs Astrobiology Institute. Mars, for example, is iron-rich and has water flowing beneath its surface. ŌĆ£If you have a system that can pick up electrons from iron and have some water, then you have all the ingredients for a conceivable metabolism,ŌĆØ said El-Naggar. Perhaps a former mine a mile underneath South Dakota wonŌĆÖt be the most surprising place that researchers find electron-eating life.”

“Types of soil in the U.S. include (clockwise from top left) the Honga, Madison, Coxville, Cowarts, Lynn Haven, Green Level, Leon, Hiwassee, Tuxekan, and Appling series. All are found in the Southeast except for the Tuxekan series, which is located in Alaska. Images courtesy of John A. Kelley, USDA Natural Resources Conservation Service”
UNDERGROUND LIFE
https://eldeveloper.github.io/cogs220
https://quantamagazine.org/imagining-data-without-division
https://quantamagazine.org/how-soil-microbes-affect-the-environment
Below Our Feet, a World of Hidden Life
by Elizabeth Svoboda┬Ā / ┬ĀJune 16, 2015
“Janet Jansson first started to wonder about the vast universe of underground life as a student at New Mexico State University in the late 1970s. A handful of soil contains about 10 billion bacteria, but at the time, earth scientists knew very little about what these microbes were and what they did. Later, as a young microbial ecologist at Stockholm University in Sweden, she started to catalog the microorganisms she collected during soil sampling trips, deciphering their genetic code so she could understand both their internal workings and how they fit into their underground habitat. As Jansson dug, though, she kept running into a problem. The main method then used to amplify and analyze stretches of DNA wasnŌĆÖt powerful enough to reveal all the workings of a single microorganism, much less an entire community of them. ŌĆ£You could get information about specific genes, but sequencing technologies were very slow,ŌĆØ said Jansson, now a division director of biological sciences at Pacific Northwest National Laboratory (PNNL) in Richland, Wash. She knew the layers of sediment she studied held a treasure trove of biological finds, but she didnŌĆÖt yet have the tools she needed to unearth them.
Then, soon after the turn of the century, new high-octane DNA sequencing methods made it possible to sequence thousands or even millions of genes almost instantly. These new, speedier methods meant researchers could easily sequence the collective genomes of the sample, known as a metagenome, for the first time. Suddenly, it was possible to scan the overall composition of habitats as diverse as stagnant bogs and frozen tundra, producing a detailed portrait of the microbial life they held. The gene and protein sequences from these wide-ranging scans ŌĆö the first of their kind ŌĆö would, once decoded, illuminate what the microbes were actually doing within each ecosystem. The data would help researchers understand how microbes capture and store carbon dioxide from the atmosphere, how they break down organic matter so that plants can access its nutrients, and how they neutralize soil toxins known to threaten human health. ŌĆ£You can just sequence everything,ŌĆØ Jansson said. ŌĆ£ThatŌĆÖs where the metagenomic approach has really been an advantage.ŌĆØ
Almost everything about JanssonŌĆÖs current surroundings is big and bold: the 600 acres of sycamore-studded PNNL campus rolling across eastern Washington, the endless blue sky she can see through her picture window, the fridge-size gene sequencing machine where her team deposits soil samples. But, as ever, what drives Jansson is the lure of the microscopic and the unseen ŌĆö the challenge of mapping the contents of the soil microbiome, a teeming global community whose functions have never been fully understood. ŌĆ£Soil,ŌĆØ Jansson said, flashing a grin, ŌĆ£is the ultimate complex system.ŌĆØ Once metagenomics began opening up a whole new subsurface world to soil scientists, Jansson found herself faced with a fresh set of challenges. The scope of this emerging field she had helped to create was immense ŌĆö a single teaspoon of soil may contain tens of thousands of species, and there are perhaps millions of species worldwide that have not yet been discovered. To tackle the staggering task of understanding their function, she formed a┬Ācollaborative venture┬Āin 2010 called the Earth Microbiome Project, along with┬ĀRob Knight┬Āof the University of California, San Diego, and┬ĀJack Gilbert┬Āof Argonne National Laboratory in Argonne, Ill. The project aims to catalog 200,000 microbe-rich samples from locations around the world. (As of last year, it had analyzed more than 30,000 samples.) But Jansson also knew that to really dig deep, she had to narrow the dizzying array of research possibilities before her to a handful that she could tackle within a lifetime.

“Janet Jansson on an expedition to Greenland”
To begin, she and her team set out to study the prairie soils that dominate much of the American heartland. Thousands of soil microbes around the roots of grasses help the Midwestern Great Prairie to store more carbon than any other region in the continental United States. By producing proteins that slice and recombine carbon dioxide molecules, these microbes work in tandem with the grasses to capture atmospheric carbon dioxide and turn it into solid, carbon-rich biological matter that gets stored underground. ThatŌĆÖs a huge boon to humanity, because the more carbon dioxide a landscape can store, the less will be left as a greenhouse gas that drives planetary warming. But since over 90 percent of all soil bacteria canŌĆÖt be grown in the lab, researchers have long been unsure just how they contribute to carbon cycling. A metagenomic analysis of these prairie microbes, Jansson thought, would help reveal the extent of their involvement in carbon storage and illuminate whether rainfall and human land cultivation change that role.
JanssonŌĆÖs team fanned out across the central third of the country to see what microbial novelties they could find. At field sites in Iowa, Kansas and Wisconsin, they took soil samples using tools called corers ŌĆö foot-long hollow tubes designed to slice through the soil. When the corer emerges, it pulls up a log-shaped sample with all its layers ŌĆö and, ideally, its microorganisms ŌĆö intact. The chunk of dirt is then preserved on dry ice and sent back to the lab. There, technicians sequence the sampleŌĆÖs DNA and RNA. Once this process is complete, project scientists have a good idea which microbial genes are contained in each sample and what biological jobs the microbes perform. If a soil sample contains bacterial genes that produce enzymes used to convert carbon dioxide from the atmosphere into solid carbon, and these genes are active, researchers can conclude that microbes within the sample are actively storing carbon.
At each site, the research team sampled native, undisturbed prairie soil, as well as soil that had been farmed for many years. The sequenced samples revealed that the native prairie dirt contained a different mix of microbes from the farmed soil, which may in part be due to fertilizers that are used during farming. ŌĆ£When we looked at those comparisons, there were strong microbial signatures, almost like a biomarker of cultivation,ŌĆØ Jansson said. She suspects that there are differences in the way microbes store carbon in native and cultivated soil, a topic she plans to explore further in future research. Meanwhile, Jansson and her team were also exploring the soils of the Arctic, one of the worldŌĆÖs fastest-changing climates. Jansson had long been curious about how these rapid temperature changes were affecting the underground microbial community, and whether these changes had any surprising side effects. ŌĆ£ThereŌĆÖs so much organic carbon trapped in permafrost,ŌĆØ she said, ŌĆ£and we donŌĆÖt really know whatŌĆÖs going to happen as the climate warms.ŌĆØ At the outset of her work, Jansson suspected that warming trends might activate bacterial processes that break down stored carbon, releasing it into the atmosphere and fueling climate change. But since she didnŌĆÖt know for sure, she decided to study how the microbial community mix varied in three distinct types of Alaskan terrain: frozen permafrost, surface soil that freezes and thaws as the seasons change, and relatively warm, waterlogged bog soil.
As she suspected, Jansson found substantial differences among the microbial communities at these locations. Overall, there were few genes and proteins in the permafrost samples. But in the freezing and thawing soil layer, sequencing showed that bacteria within the soil samples were producing some intriguing proteins, including enzymes that snip long chains of carbon molecules, like cellulose from plants, into shorter, simpler sugar compounds that the bacteria can use as fuel. When this happens, previously ŌĆ£locked-downŌĆØ carbon is released back into the atmosphere. ŌĆ£When [the soil] thaws,ŌĆØ Jansson said, ŌĆ£it starts to transition more into decomposition.ŌĆØ In warmer soils, in other words, bacterial carbon breakdown processes start to show up clearly in sequencing data. So just as some observers have feared, warming temperatures could release formerly inert carbon from the soil. This raises the worrying prospect of runaway carbon release as temperatures continue to rise.

“Soil cores are transported back to the lab, where researchers can analyze the dense microbial populations.”
Most strikingly, the warmest soil sample in JanssonŌĆÖs study ŌĆö the spongy bog soil ŌĆö revealed an array of microbial genes and proteins involved in the production of methane, a greenhouse gas more than 20 times as potent as carbon dioxide. One such protein was methyl-coenzyme M reductase, which is involved in transforming carbon dioxide into methane. This finding might imply that warming trends will drive local microbes to produce larger amounts of methane. Next, Jansson plans to investigate whether rapid thaws affect soil microbe populations differently from more gradual thaws, since both will likely become commonplace as the planet warms. Soil microbes are not just carbon processors, as the work of JanssonŌĆÖs colleagues reveals. Not only do the vast microbial communities underfoot affect air quality and global temperatures, they can also affect the taste and quality of the food we grow.┬ĀThomas Mitchell-Olds┬Āof Duke University wanted to see if the microbial populations in and around plantsŌĆÖ roots could affect the way the plants matured. He and his student┬ĀMaggie Wagner┬Ātook soil samples from four different collecting sites in rural Idaho and isolated microbes from each sample. They then inoculated soil-filled pots with these four microbial samples and planted a common strain of mustard plant,┬ĀBoechera stricta, in them. Certain types of microbes in the soil seemed to speed plant flowering time, the team found, while others, such as members of the┬ĀProteobacteria┬Āphylum, slowed it down.
The study underscored just how necessary microbial activity is to plant health and productivity. ŌĆ£Soil microbes had already been linked to drought tolerance, growth rate and other aspects of plant performance. Our experiment added flowering time to this list,ŌĆØ Mitchell-Olds said. ŌĆ£The potential for soil microbes to tweak flowering time ŌĆö whether to improve yield, buffer against climate change, or both ŌĆö is intriguing.ŌĆØ Such a prospect might interest not just farmers looking to increase their bounty, but also, say, wine growers in CaliforniaŌĆÖs Napa Valley, who know how dramatically bloom time can affect the development of grapes and the taste of the resulting vintage. The famed terroir of places like Napa,┬Āin fact, may arise from thousands of microbes humming along in unseen harmony.
Other sequencing studies have illustrated the critical role soil microbes play in breaking down pollutants. When scientists at the University of Delhi in India took soil samples from a pesticide dump and compared them with samples from a cleaner control site, they reported that the soil from the waste site contained a higher concentration of gene sequences from certain bacterial groups, such as Pseudomonas,┬ĀNovosphingobium┬Āand┬ĀSphingomonas, that are known to degrade common pesticides like hexachlorocyclohexane. Microbes, it seems, can adapt to help fouled landscapes recover, which presents the exciting possibility of deploying the bugs in bioremediation efforts. ŌĆ£Soils are a buffer against all kinds of insults to our ecosystem,ŌĆØ said┬ĀVanessa Bailey, a microbiologist who works closely with Jansson at PNNL.
The researchers working on the Great Prairie project have managed to compile about 1.8 trillion bases of DNA data, but Jansson thinks those data describe only a fraction of the microbial communities in prairie soil, given that the soil contains between 1 billion and 10 billion individual cells per gram. ŌĆ£WeŌĆÖre still at the discovery phase,ŌĆØ she said. ŌĆ£We have a lot to do just to assemble a soil metagenome.ŌĆØ She and other scientists are trying various techniques to streamline the gene analysis process, such as discarding less common gene sequences in a particular sample in order to focus on more prevalent ones. The more Jansson and her colleagues are able to learn about previously unknown communities of microbes, the better theyŌĆÖll be able to predict how these communities will react to different conditions ŌĆö droughts, warmer weather or floods, just to kick off the list. They hope to build these predictions into computer models that would illustrate what microbial activity a given environmental change might create, as well as the expected results of that activity. Such models could help environmental planners cultivate microbial mixes that achieve a desired end ŌĆö which could be soil that locks up gigatons of atmospheric carbon, or that sloughs off pollution with ease, or that yields the kinds of grapes vintners dream about. ŌĆ£By having knowledge of the microbes that are present in the best-case scenario,ŌĆØ Jansson said, ŌĆ£you can tweak the system to optimize that combination of microbes.ŌĆØ
But achieving the right microbial balance isnŌĆÖt always as simple as inoculating soil with certain strains of bacteria. ŌĆ£If the conditions are favorable for the microbes you want to flourish, they will flourish,ŌĆØ Jansson said. ŌĆ£If you seed and the conditions arenŌĆÖt favorable, they will just die.ŌĆØ The ideal approach, then, will often be to engineer the kind of landscape that beneficial microbes naturally flock to. In warming areas that grow boggier every year, this might mean ensuring that there is enough oxygen-rich moving water, which would make the area less hospitable to anaerobic microbes that belch large quantities of methane.

“Two views of the carbonate chimneys at the Point Dume methane seep off southern California are covered with colorful microbial mats and permeated by methane-eating microbes.”┬Ā
In cultivated areas of the Great Prairie, farmers might choose fertilizers that preserve natural microbial diversity, yielding plentiful food crops and maximizing carbon capture from the atmosphere. Still, insights like these are gradual and hard-won, and Jansson knows future scientists will need to take up the cause of discovering all the ways soil microbes affect the rest of the planet. For now, Jansson relishes playing the role of soil surveyor, mapping the variety and breadth of microbial communities so others can someday benefit from the knowledge. ŌĆ£We know more about the movement of celestial bodies,ŌĆØ Leonardo da Vinci once mused, ŌĆ£than about the soil underfoot.ŌĆØ More than 500 years later, Jansson wants to be the scientist who finally proves him wrong.”
PREVIOUSLY
DEEP HOT BIOSPHERE
https://spectrevision.net/2020/01/27/deep-hot-biosphere/
LAZARUS MICROBES
https://spectrevision.net/2017/05/22/resurrection-ecology/
ASTROBIOLOGY
https://spectrevision.net/2020/09/18/biosignatures-on-venus/
ELECTROACTIVE MICROBES
https://spectrevision.net/2019/07/04/live-wires/
RADIATION RESISTANT MICROBES
https://spectrevision.net/2018/01/30/radiation-resistant-microbes/
GEOACTIVE MICROBES
https://spectrevision.net/2020/10/01/radical-geomycology/