GENETIC RESCUE
https://reviverestore.org/what-we-do/genetic-rescue-toolkit
https://nationalgeographic.com/podcasts/episode-8-frozen-zoo
https://hackaday.com/cloning-frozen-zoo-seed-bank-for-animals
Like a Seed Bank but for Animals
by Lewin Day / March 4, 2021
“Seed banks are facilities of great value to biodiversity and agriculture around the world. These facilities are used to house stocks of seeds of a wide variety, helping to maintain genetic diversity and avoid the permanent loss of various plant species. While there are some challenges, the basic requirements to run a simple seed bank are to keep a selection of seeds at low temperature and humidity to maximise their viable lifespan.
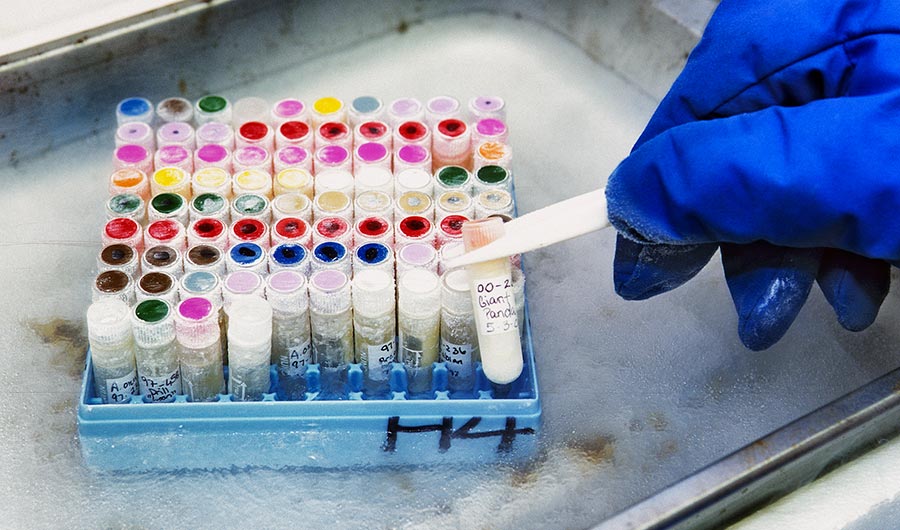
When it comes to animals, things become more difficult — one can’t simply plant an old seed in the ground and grow a fresh new meerkat, for example. Preservation of animal genetic material poses its own unique set of challenges — ones that the San Diego “Frozen Zoo” don’t shy away from. They’ve recently shown the viability of the program with the healthy birth of a ferret cloned from an animal that died in 1988.
There are less than a dozen frozen zoos around the world, with the first being established at San Diego Zoo by Kurt Benirschke in 1972. While the available biological technology was limited at the time, it was hoped that by cryopreserving samples of animal tissues and reproductive material, they might later be used for research or reproduction purposes. The idea has since spread, with a smattering of other facilities opening up around the world. Such facilities necessarily store a wide variety of material, depending on the species in question. Obviously, viable gametes, or reproductive cells, are of high priority. Eggs and sperm cells from sexually mature animals can readily be secured from both live and deceased specimens, and used to produce embryos for implantation.

“Dolly and her surrogate mother”
Alternatively, fluids such as blood or milk may be saved, as well as muscle tissue, bone, hair or skin samples. With cloning techniques pioneered in the 1990s, DNA extracted from these non-reproductive cells can be inserted into an egg with its nucleus removed. This egg can then be implanted in a surrogate mother like any other embryo, and the pregnancy carried to term. With in vitro fertilization (IVF) techniques in their infancy in the 1970s and cloning a distant blip on the horizon, Dr. Benirschke’s decision to establish the first frozen zoo at the time shows considerable foresight.

“first clone of a US endangered species”
With decades of advancements in genetic sequencing and techniques like in vitro fertilization now available, these libraries of genetic material are starting to bear fruit. Although other species have already been successfully cloned, this is the first clone of a US endangered species — a black-footed ferret by the name of Elizabeth Ann. Born to a domestic ferret serving as a foster mother on December 10th of last year, her genes are a duplicate of a ferret named Willa who passed away in 1988 and was frozen at the San Diego facility. The black-footed ferret has long been a focus of conservationists, who have been working to re-establish the species since it was thought to be extinct in the mid-20th century. When a dead specimen was found on a ranch in the 1980s, breeding work began in earnest, with thousands of ferrets reintroduced into the wild.
As for Elizabeth Ann and any potential future clones, however, there aren’t yet plans to release them from captivity. Both breeding programs and cloning experiments highlight a limitation of this work, however. Genetic diversity is key to maintaining a thriving population over many generations, but the ferrets bred as part of the program all trace their lineage to just 7 individuals. Similarly, a cloning program can produce theoretically unlimited offspring from a single DNA sample, but inbreeding depression will make such a cohort unlikely to thrive in the long term. Thus, any frozen zoo aiming to serve as a potential backup against possible extinctions needs to collect as broad a spectrum of genetic samples as possible.

“Kurt is the world’s first Przewalski’s horse clone, born to a domestic horse surrogate mother. It’s hoped that Kurt will grow up to breed with others of his species, increasing genetic diversity of the herd.”
The San Diego facility has had other success stories, too. In partnership with Viagen, a company perhaps best known for offering cloning services for domestic pets, scientists were able to clone Kurt, a Przewalski’s Horse, from forty-year old frozen skin samples. Similarly to the methods used to create Elizabeth Ann, Kurt’s genetic material was used to fertilise an egg which was then implanted into a domestic horse serving as a surrogate mother. Sequencing revealed the donor tissue featured many unique genes not found in the currently-alive population, all of which come from lines of just 12 former individuals. It’s hoped that when Kurt comes of age, breeding with others will significantly increase the genetic diversity of the endangered species.
The need for similar species to carry pregnancies means that it’s unlikely we’ll see frozen zoos churning out wooly mammoths or Tasmanian tigers for some time yet. Having the genetic material alone isn’t enough; a suitably close living relation is key, along with the aforementioned need for genetic diversity if repopulation is the goal. However, the technologies and techniques that have been developed will be crucial to maintaining biodiversity of existing species well into the future, especially given that habitat destruction and other existential threats remain around the world. And, as science continues to progress, it’s likely that frozen zoos will be the first to invite you to see their new dodo exhibit in the future.”
CRYOBIOLOGY
https://societyforcryobiology.org
https://science.sandiegozoo.org/frozen-zoo
https://insidescience.org/frozen-zoos-put-endangered-species-ice
How Caves & Frozen Zoos Are Putting Endangered Species on Ice
by Katharine Gammon / August 1, 2019
“In 1975, medical doctor Kurt Benirschke founded the Center for the Reproduction of Endangered Species with the goal of using molecular genetics tools to save endangered species. In the corner of the modest lab, which contained a freezer with liquid nitrogen to bank cells, Benirschke hung a poster: “You must collect things for reasons you don’t yet understand.” That credo holds true for scientists in cryobiology today. Researchers are in a race against time to bank the tissues and cells of creatures and plants disappearing from our world. They don’t necessarily know what they’ll be able to do with these genetic libraries in the future, but to lose them would be unthinkable in the midst of a global extinction crisis.
At the Society for Cryobiology annual meeting in San Diego last week, Oliver Ryder spoke about how precious and irreplaceable biobanks are. Ryder directs the San Diego Zoo’s “Frozen Zoo” program, the largest animal cryobank in the world. “Being able to successfully preserve samples is the opportunity of our time, and one that will impact the future,” he told the audience of scientists gathered at the conference. The repository holds nearly 10,000 cell cultures of 1,000 different species. They’re mostly mammals, but the cryobank also includes samples from birds, reptiles and amphibians. The trick with cryopreserving anything is to avoid forming ice crystals as the samples drop in temperature to minus 196 degrees Celsius in liquid nitrogen. That temperature arrests all biological activity, including the degradation of genetic material. Depending on the sample, scientists might use different techniques to prevent ice crystals from developing, but the most common method is to treat the cells with dimethyl sulfoxide. It basically turns the water-based samples into glass.
There is a lot of information inside those banked cells. They can contribute to a better scientific understanding of evolution and how species are connected, but also help with studies on aging, genetics and genealogy. They can also help scientists learn more about animal health research and practice, and stem cells. In addition, one of the primary goals is to help add genetic diversity to threatened or endangered populations that are at risk of being inbred. “If you can flow in genes from another population, the recipient population can show a dramatic increase of fitness,” said Ryder. Scientists could potentially use banked cells to clone long-dead animals that would then add to the genetic diversity of the distressed population.
One of the species that the Frozen Zoo aims to rescue is the northern white rhino. As of today, the only living females, both in Kenya, are too old to reproduce. But the Frozen Zoo scientists have banked 12 cell cultures over the years. Once they develop a reliable technique, they’d like to thaw the cells, turn them into induced pluripotent stem cells, and make egg and sperm in order to raise new rhino babies. Some species are easier to freeze than others. Scientists have been successful freezing mammal and bird cells, but have had a harder time with amphibian cells, succeeding less than 1% of the time. Other researchers presenting at the conference were attempting even more ambitious fauna: coral reefs, mollusks, snakes, fish and eels, among others.
It’s not easy to get access to endangered creatures. Often, the researchers collect a pea-sized amount of tissue from a living animal during an exam, or gather the notch of skin that zookeepers remove from an animal’s ear to identify it. They also take samples during autopsy examinations, which all zoo animals undergo, said Marlys Houck, the Frozen Zoo’s curator. Although they’d like to be able to save every species before the last individuals die, the end goal of the cryobank is often to gather data, she said. “Sometimes it’s just saving some information about that species, so we’d have a living cell that you can look at the genome and see how it’s related to other species. We don’t have that for the dodo or the passenger pigeon.”
There’s another famous cryobank, the Svalbard seed vault. Inside this vast mountain cave tunnel between Norway and the North Pole are millions of the world’s important crop seeds, sealed in specially designed three-ply foil packages placed in sealed boxes and stored on shelves inside the vault. Svalbard is a frosty minus 18 degrees Celsius, held steady by the surrounding permafrost. More than 820,000 seed samples are stored in the vault. But the problem, says Bart Panis, a senior researcher with the Leuven, Belgium-based nonprofit Bioversity International, is that not all seeds can be stored at that temperature. Crops that produce a so-called recalcitrant seed — including popular plants like avocado, mango, rubber and cocoa — can’t be stored at this temperature without losing viability for regeneration. In addition, for sterile crops like banana — which lack seeds — seed conservation is not an option.
For these crops, other methods are needed. Panis described the method for genebanking bananas and their close relatives. Together, they are considered a staple food that provides the basis for nutrition for up to 1 billion people each day. Bioversity has amassed the largest collection of banana germplasm — the tissue from which new plants can be born. This is how it works: The researchers grow the bananas in test tubes in a temperate environment, about 15 degrees C — about 10 degrees colder than their typical habitat. They also clip a tiny part of the plant’s growing point (known as the maristem) and freeze that to minus 196 degrees, which preserves the plants indefinitely, but allows the plants to be revived again when needed.
Panis and the other researchers have amassed a reference collection of more than 1,500 banana samples. That’s the largest number of banana samples anywhere in the world. They also have a backup location in Montpelier, France, in case something happens to the building in Leuven. San Diego’s Frozen Zoo has a backup site for frozen sample about 30 miles from their main location. Ryder says that cryopreservation may actually change the way humanity thinks about conservation. “Because try as we might, we are losing species. And if we don’t acknowledge our lack of prowess as a civilization in saving species, we are always going to be banking the last one,” he said. “But if we start to anticipate that not everything is going to be saved, but we could collect biodiversity, we may be able to offer the future a richer legacy.” For some species, cell-based technology may be the only way to save them in perpetuity, and we need to gather and preserve samples as soon as possible, said Ryder. “You can only save species that you bank.”
PREVIOUSLY
GUARDED by POLAR BEARS, for NOW
https://spectrevision.net/2006/06/20/guarded-by-polar-bears-for-now/
RESURRECTION ECOLOGY
https://spectrevision.net/2017/05/22/resurrection-ecology/
DE-EXTINCTION
https://spectrevision.net/2013/05/09/de-extinction/