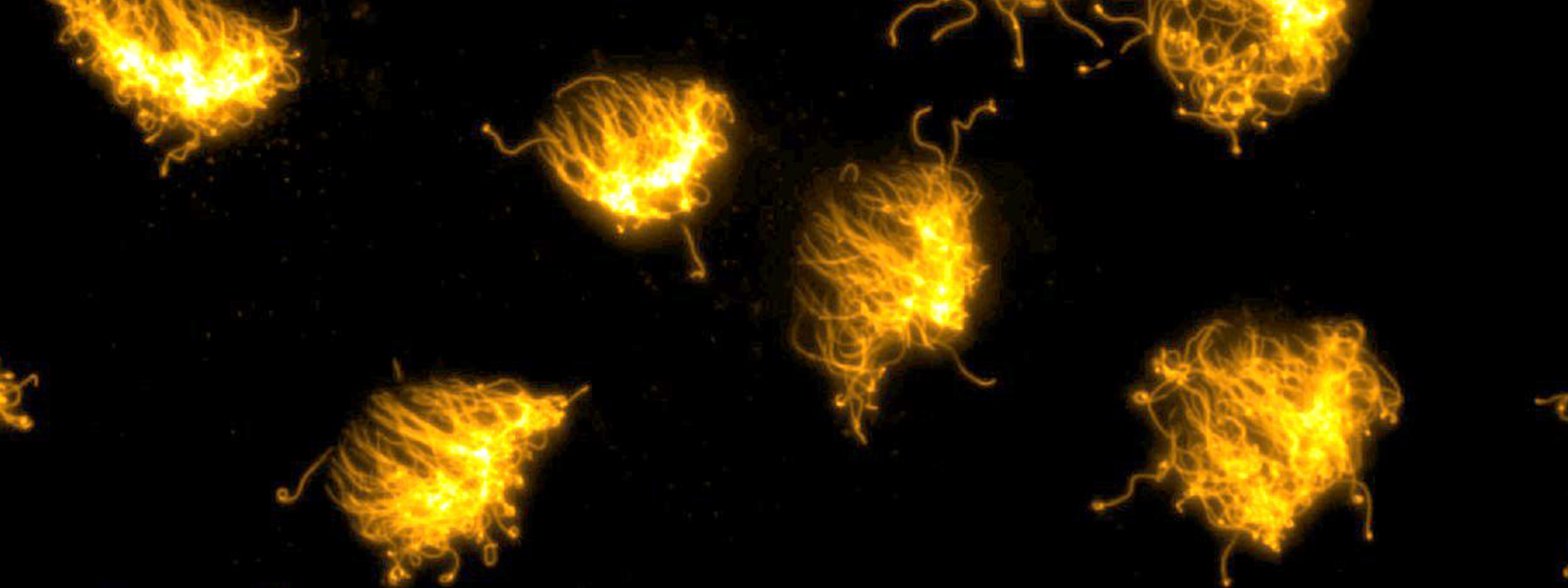
XENOBOTS
https://krorgs.github.io/videos
https://en.wikipedia.org/wiki/Xenobot
https://usatoday.com/living-robots-reproduce-artificial-intelligence
Scientists made tiny xenobots out of frog cells. Now they say those robots can reproduce.
by Jordan Mendoza / 11/30/21
“Life finds a way, and the same goes for even robots, according to a group of scientists who say the first living robotic life forms can reproduce. In January 2020, a team of scientists from the University of Vermont, Tufts University and Harvard University took stem cells from African clawed frog embryos and formed them into tiny living creatures called xenobots. The xenobots, which are less than 0.04 inches wide, were able to move on their own, communicate amongst each other and heal themselves from an injury, making them the first-ever living robots. But over one year later, the computer-designed creatures have begun to do “something that’s never been observed before.”
Our third xenobots paper was published today in PNAS.
In a nutshell: they can now self-replicate.
w/ @DougBlackiston, @drmichaellevin, @DoctorJosh
More info: https://t.co/ySLBHeaGlL pic.twitter.com/vTvS2E3PoG
— Sam Kriegman (@Kriegmerica) November 29, 2021
What the team of scientists discovered was the xenobots would move around their environment and find single cells. They would gather hundreds of these cells at once and then assemble an offspring inside their mouth. A few days later, the offspring became a new xenobot that functioned as the others. The group published their findings in the peer-reviewed journal PNAS on Monday. “This is profound,” Michael Levin, director of the Allen Discovery Center at Tufts University and co-leader of the new research, said in a statement. “These cells have the genome of a frog, but, freed from becoming tadpoles, they use their collective intelligence, a plasticity, to do something astounding.” Sam Kriegman, a post-doctoral researcher at Tufts and Harvard and the lead author of the study, said what makes the discovery so remarkable is the xenobots reproduced in a way that most animals wouldn’t.
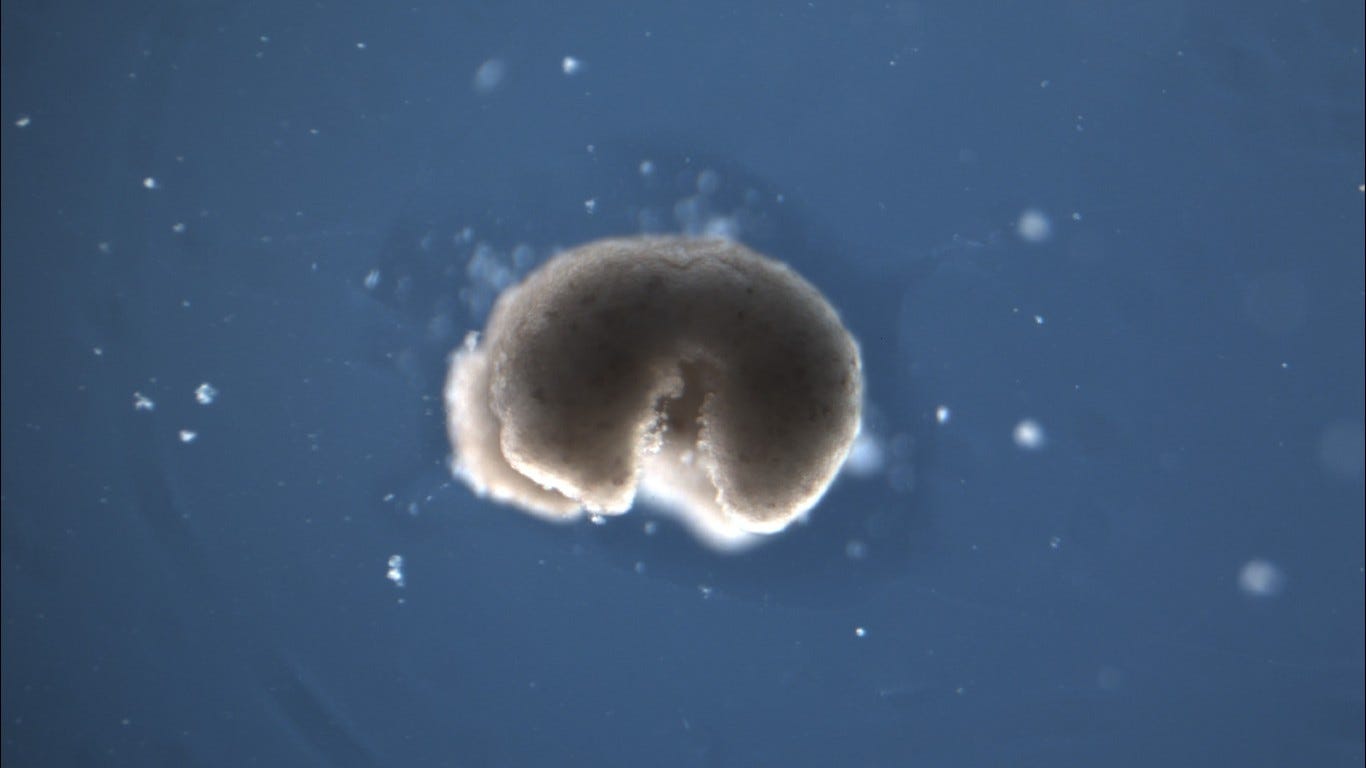
“Xenobots are small biological machines, or living robots, created from frog cells by biologists and computer scientists from Tufts University and University of Vermont”
When the scientists created the xenobots, they stripped all frog characteristics from them, meaning they can’t replicate through creating tadpoles. The original design of the bots wasn’t successful in reproducing, so the group used artificial intelligence to help decide what would be the best design for them to reproduce. The best body shape wound up looking very similar to the iconic video game character, Pac-Man. When they took the Pac-Man shape and put it in a petri dish, it began the reproduction process and was successful. This process of reproduction, known as kinematic replication, is very common, but only in molecules. “No animal or plant known to science replicates in this way,” Kriegman said.
While this may sound like the beginning of the plot to a “Terminator” movie, Joshua Bongard, a computer scientist and robotics expert at the University of Vermont and study co-author, says there is no worry that this may lead to the end of human civilization. The xenobots are small, only live in a laboratory and can be easily killed. Bongard said artificial intelligence being able to develop the ability for xenobots to reproduce may be beneficial to any problems living creatures face, such as birth defects, diseases and cancer. “All of these different problems are here because we don’t know how to predict and control what groups of cells are going to build. Xenobots are a new platform for teaching us. “We found Xenobots that walk. We found Xenobots that swim. And now, in this study, we’ve found Xenobots that kinematically replicate. What else is out there?” Bongard said.”
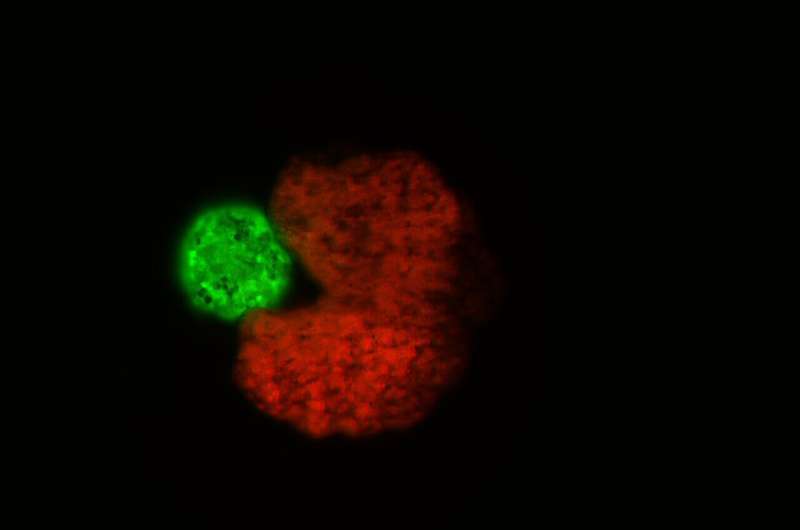
“An AI-designed, Pac-Man-shaped “parent” organism (in red) beside stem cells that have been compressed into a ball—the “offspring” (green)”
HOW to MAKE FROGS without TADPOLES
https://www.freethink.com/science/xenobots
https://phys.org/bot-beast-scientists-programmable
https://techxplore.com/news/2021-11-xenobots-team-robots.html
Team builds first living robots that can reproduce
by Joshua Brown / November 29, 2021
“To persist, life must reproduce. Over billions of years, organisms have evolved many ways of replicating, from budding plants to sexual animals to invading viruses. Now scientists have discovered an entirely new form of biological reproduction—and applied their discovery to create the first-ever, self-replicating living robots. The same team that built the first living robots (“Xenobots,” assembled from frog cells—reported in 2020) has discovered that these computer-designed and hand-assembled organisms can swim out into their tiny dish, find single cells, gather hundreds of them together, and assemble “baby” Xenobots inside their Pac-Man-shaped “mouth”—that, a few days later, become new Xenobots that look and move just like themselves. And then these new Xenobots can go out, find cells, and build copies of themselves. Again and again. “With the right design—they will spontaneously self-replicate,” says Joshua Bongard, a computer scientist and robotics expert at the University of Vermont who co-led the new research. The results of the new research were published November 29, 2021, in the Proceedings of the National Academy of Sciences.

“Spontaneous kinematic self-replication.”
In a Xenopus laevis frog, these embryonic cells would develop into skin. “They would be sitting on the outside of a tadpole, keeping out pathogens and redistributing mucus,” says Michael Levin, a professor of biology and director of the Allen Discovery Center at Tufts University and co-leader of the new research. “But we’re putting them into a novel context. We’re giving them a chance to reimagine their multicellularity.” And what they imagine is something far different than skin. “People have thought for quite a long time that we’ve worked out all the ways that life can reproduce or replicate. But this is something that’s never been observed before,” says co-author Douglas Blackiston, the senior scientist at Tufts University who assembled the Xenobot “parents” and developed the biological portion of the new study. “This is profound,” says Levin. “These cells have the genome of a frog, but, freed from becoming tadpoles, they use their collective intelligence, a plasticity, to do something astounding.” In earlier experiments, the scientists were amazed that Xenobots could be designed to achieve simple tasks. Now they are stunned that these biological objects—a computer-designed collection of cells—will spontaneously replicate. “We have the full, unaltered frog genome,” says Levin, “but it gave no hint that these cells can work together on this new task,” of gathering and then compressing separated cells into working self-copies.
Very nice, from @CejkovaJitka and colleagues – https://t.co/50h923T4oh – right on, droplets as a platform for liquid *robots*. Indeed!
— Michael Levin (@drmichaellevin) December 11, 2021
“These are frog cells replicating in a way that is very different from how frogs do it. No animal or plant known to science replicates in this way,” says Sam Kriegman, the lead author on the new study, who completed his Ph.D. in Bongard’s lab at UVM and is now a post-doctoral researcher at Tuft’s Allen Center and Harvard University’s Wyss Institute for Biologically Inspired Engineering. On its own, the Xenobot parent, made of some 3,000 cells, forms a sphere. “These can make children but then the system normally dies out after that. It’s very hard, actually, to get the system to keep reproducing,” says Kriegman. But with an artificial intelligence program working on the Deep Green supercomputer cluster at UVM’s Vermont Advanced Computing Core, an evolutionary algorithm was able to test billions of body shapes in simulation—triangles, squares, pyramids, starfish—to find ones that allowed the cells to be more effective at the motion-based “kinematic” replication reported in the new research. “We asked the supercomputer at UVM to figure out how to adjust the shape of the initial parents, and the AI came up with some strange designs after months of chugging away, including one that resembled Pac-Man,” says Kriegman. “It’s very non-intuitive. It looks very simple, but it’s not something a human engineer would come up with. Why one tiny mouth? Why not five? We sent the results to Doug and he built these Pac-Man-shaped parent Xenobots. Then those parents built children, who built grandchildren, who built great-grandchildren, who built great-great-grandchildren.” In other words, the right design greatly extended the number of generations.

“Amplifying kinematic self-replication. Due to surface tension, reconfigurable organisms naturally develop into ciliated spheroids, but they can be sculpted into nonspheroidal morphologies manually during development to realize more complex body shapes.”
Kinematic replication is well-known at the level of molecules—but it has never been observed before at the scale of whole cells or organisms. “We’ve discovered that there is this previously unknown space within organisms, or living systems, and it’s a vast space,” says Bongard, a professor in UVM’s College of Engineering and Mathematical Sciences. “How do we then go about exploring that space? We found Xenobots that walk. We found Xenobots that swim. And now, in this study, we’ve found Xenobots that kinematically replicate. What else is out there?” Or, as the scientists write in the Proceedings of the National Academy of Sciences study: “life harbors surprising behaviors just below the surface, waiting to be uncovered.” Some people may find this exhilarating. Others may react with concern, or even terror, to the notion of a self-replicating biotechnology. For the team of scientists, the goal is deeper understanding. “We are working to understand this property: replication. The world and technologies are rapidly changing. It’s important, for society as a whole, that we study and understand how this works,” says Bongard.
These millimeter-sized living machines, entirely contained in a laboratory, easily extinguished, and vetted by federal, state and institutional ethics experts, “are not what keep me awake at night. What presents risk is the next pandemic; accelerating ecosystem damage from pollution; intensifying threats from climate change,” says UVM’s Bongard. “This is an ideal system in which to study self-replicating systems. We have a moral imperative to understand the conditions under which we can control it, direct it, douse it, exaggerate it.” Bongard points to the COVID epidemic and the hunt for a vaccine. “The speed at which we can produce solutions matters deeply. If we can develop technologies, learning from Xenobots, where we can quickly tell the AI,: ‘We need a biological tool that does X and Y and suppresses Z,’ —that could be very beneficial. Today, that takes an exceedingly long time.”
The team aims to accelerate how quickly people can go from identifying a problem to generating solutions—”like deploying living machines to pull microplastics out of waterways or build new medicines,” Bongard says. “We need to create technological solutions that grow at the same rate as the challenges we face,” Bongard says. And the team sees promise in the research for advancements toward regenerative medicine. “If we knew how to tell collections of cells to do what we wanted them to do, ultimately, that’s regenerative medicine—that’s the solution to traumatic injury, birth defects, cancer, and aging,” says Levin. “All of these different problems are here because we don’t know how to predict and control what groups of cells are going to build. Xenobots are a new platform for teaching us.”
More information: Kinematic self-replication in reconfigurable organisms, Proceedings of the National Academy of Sciences (2021). DOI: 10.1073/pnas.2112672118

“Xenobots exhibit cooperative swarm activity, in this case working together to gather piles of tiny particles”
SPONTANEOUS SELF-REPLICATION
https://direct.mit.edu/Droplets-As-Liquid-Robots
https://news.mit.edu/designing-training-intelligent-soft-robots
https://techxplore.com/news/2021-03-scientists-robots.html
Scientists create the next generation of living robots
by Tufts University / March 31, 2021
“Last year, a team of biologists and computer scientists from Tufts University and the University of Vermont (UVM) created novel, tiny self-healing biological machines from frog cells called “Xenobots” that could move around, push a payload, and even exhibit collective behavior in the presence of a swarm of other Xenobots. Get ready for Xenobots 2.0. The same team has now created life forms that self-assemble a body from single cells, do not require muscle cells to move, and even demonstrate the capability of recordable memory. The new generation Xenobots also move faster, navigate different environments, and have longer lifespans than the first edition, and they still have the ability to work together in groups and heal themselves if damaged.
The results of the new research were published today in Science Robotics. Compared to Xenobots 1.0, in which the millimeter-sized automatons were constructed in a “top down” approach by manual placement of tissue and surgical shaping of frog skin and cardiac cells to produce motion, the next version of Xenobots takes a “bottom up” approach. The biologists at Tufts took stem cells from embryos of the African frog Xenopus laevis (hence the name “Xenobots”) and allowed them to self-assemble and grow into spheroids, where some of the cells after a few days differentiated to produce cilia—tiny hair-like projections that move back and forth or rotate in a specific way. Instead of using manually sculpted cardiac cells whose natural rhythmic contractions allowed the original Xenobots to scuttle around, cilia give the new spheroidal bots “legs” to move them rapidly across a surface.
Robots that evolve like animals: https://t.co/kaiO7zAhHv pic.twitter.com/dWlBHVzzvq
— MIT CSAIL (@MIT_CSAIL) December 8, 2021
In a frog, or human for that matter, cilia would normally be found on mucous surfaces, like in the lungs, to help push out pathogens and other foreign material. On the Xenobots, they are repurposed to provide rapid locomotion. “We are witnessing the remarkable plasticity of cellular collectives, which build a rudimentary new ‘body’ that is quite distinct from their default—in this case, a frog—despite having a completely normal genome,” said Michael Levin, Distinguished Professor of Biology and director of the Allen Discovery Center at Tufts University, and corresponding author of the study. “In a frog embryo, cells cooperate to create a tadpole. Here, removed from that context, we see that cells can re-purpose their genetically encoded hardware, like cilia, for new functions such as locomotion. It is amazing that cells can spontaneously take on new roles and create new body plans and behaviors without long periods of evolutionary selection for those features.”
“In a way, the Xenobots are constructed much like a traditional robot. Only we use cells and tissues rather than artificial components to build the shape and create predictable behavior.” said senior scientist Doug Blackiston, who co-first authored the study with research technician Emma Lederer. “On the biology end, this approach is helping us understand how cells communicate as they interact with one another during development, and how we might better control those interactions.” While the Tufts scientists created the physical organisms, scientists at UVM were busy running computer simulations that modeled different shapes of the Xenobots to see if they might exhibit different behaviors, both individually and in groups. Using the Deep Green supercomputer cluster at UVM’s Vermont Advanced Computing Core, the team, led by computer scientists and robotics experts Josh Bongard and under hundreds of thousands of random environmental conditions using an evolutionary algorithm. These simulations were used to identify Xenobots most able to work together in swarms to gather large piles of debris in a field of particles. “We know the task, but it’s not at all obvious—for people—what a successful design should look like. That’s where the supercomputer comes in and searches over the space of all possible Xenobot swarms to find the swarm that does the job best,” says Bongard. “We want Xenobots to do useful work. Right now we’re giving them simple tasks, but ultimately we’re aiming for a new kind of living tool that could, for example, clean up microplastics in the ocean or contaminants in soil.”
It turns out, the new Xenobots are much faster and better at tasks such as garbage collection than last year’s model, working together in a swarm to sweep through a petri dish and gather larger piles of iron oxide particles. They can also cover large flat surfaces, or travel through narrow capillaries.These studies also suggest that the in silico simulations could in the future optimize additional features of biological bots for more complex behaviors. One important feature added in the Xenobot upgrade is the ability to record information. A central feature of robotics is the ability to record memory and use that information to modify the robot’s actions and behavior. With that in mind, the Tufts scientists engineered the Xenobots with a read/write capability to record one bit of information, using a fluorescent reporter protein called EosFP, which normally glows green. However, when exposed to light at 390nm wavelength, the protein emits red light instead. The cells of the frog embryos were injected with messenger RNA coding for the EosFP protein before stem cells were excised to create the Xenobots. The mature Xenobots now have a built-in fluorescent switch which can record exposure to blue light around 390nm.
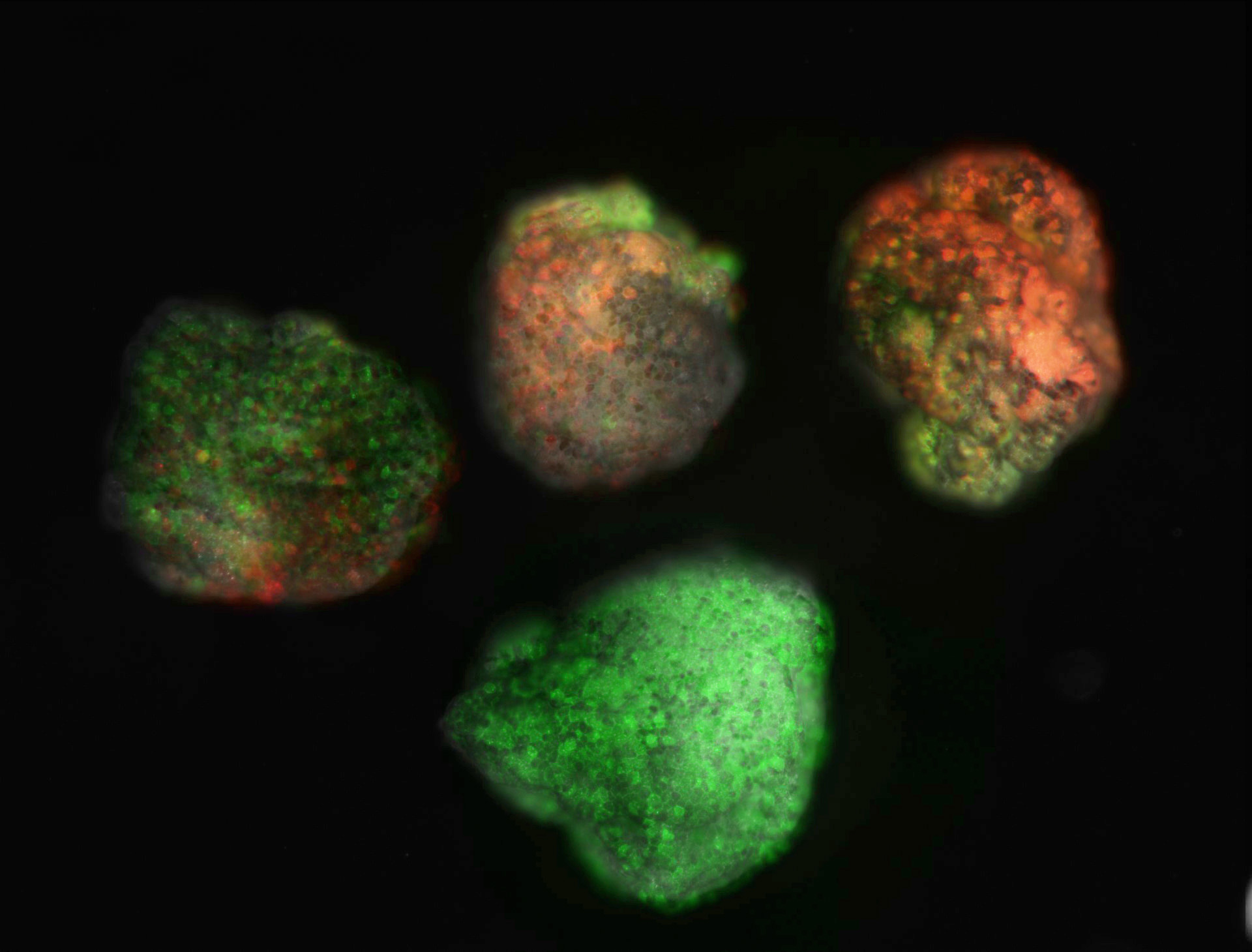
“Armed with a special protein that changes color from green to red, the next generation Xenobots could keep a simple record of their exposure to blue light”
The researchers tested the memory function by allowing 10 Xenobots to swim around a surface on which one spot is illuminated with a beam of 390nm light. After two hours, they found that three bots emitted red light. The rest remained their original green, effectively recording the “travel experience” of the bots. This proof of principle of molecular memory could be extended in the future to detect and record not only light but also the presence of radioactive contamination, chemical pollutants, drugs, or a disease condition. Further engineering of the memory function could enable the recording of multiple stimuli (more bits of information) or allow the bots to release compounds or change behavior upon sensation of stimuli. “When we bring in more capabilities to the bots, we can use the computer simulations to design them with more complex behaviors and the ability to carry out more elaborate tasks,” said Bongard. “We could potentially design them not only to report conditions in their environment but also to modify and repair conditions in their environment.”
“The biological materials we are using have many features we would like to someday implement in the bots—cells can act like sensors, motors for movement, communication and computation networks, and recording devices to store information,” said Levin. “One thing the Xenobots and future versions of biological bots can do that their metal and plastic counterparts have difficulty doing is constructing their own body plan as the cells grow and mature, and then repairing and restoring themselves if they become damaged. Healing is a natural feature of living organisms, and it is preserved in Xenobot biology.” The new Xenobots were remarkably adept at healing and would close the majority of a severe full-length laceration half their thickness within 5 minutes of the injury.
All injured bots were able to ultimately heal the wound, restore their shape and continue their work as before. Another advantage of a biological robot, Levin adds, is metabolism. Unlike metal and plastic robots, the cells in a biological robot can absorb and break down chemicals and work like tiny factories synthesizing and excreting chemicals and proteins. The whole field of synthetic biology—which has largely focused on reprogramming single celled organisms to produce useful molecules—can now be exploited in these multicellular creatures. Like the original Xenobots, the upgraded bots can survive up to ten days on their embryonic energy stores and run their tasks without additional energy sources, but they can also carry on at full speed for many months if kept in a “soup” of nutrients.
An engaging description of the biological bots and what we can learn from them is presented in a TED talk by Michael Levin (go.ted.com/michaellevin). In his TED Talk, professor Levin describes not only the remarkable potential for tiny biological robots to carry out useful tasks in the environment or potentially in therapeutic applications, but he also points out what may be the most valuable benefit of this research—using the bots to understand how individual cells come together, communicate, and specialize to create a larger organism, as they do in nature to create a frog or human. It’s a new model system that can provide a foundation for regenerative medicine. Xenobots and their successors may also provide insight into how multicellular organisms arose from ancient single celled organisms, and the origins of information processing, decision making and cognition in biological organisms. Recognizing the tremendous future for this technology, Tufts University and the University of Vermont have established the Institute for Computer Designed Organisms (ICDO), to be formally launched in the coming months, which will pull together resources from each university and outside sources to create living robots with increasingly sophisticated capabilities.”
More information: D. Blackiston el al., “A cellular platform for the development of synthetic living machines,” Science Robotics (2021). http://robotics.sciencemag.org/lookup/doi/10.1126/scirobotics.abf1571
BETTER than FROGS
https://uvm.edu/team-builds-first-living-robots-can-reproduce
https://scitechdaily.com/scientists-build-first-living-robots-that-can-reproduce
https://techxplore.com/team-robots
by University of Vermont / January 13, 2020
“A book is made of wood. But it is not a tree. The dead cells have been repurposed to serve another need. Now a team of scientists has repurposed living cells—scraped from frog embryos—and assembled them into entirely new life-forms. These millimeter-wide “xenobots” can move toward a target, perhaps pick up a payload (like a medicine that needs to be carried to a specific place inside a patient)—and heal themselves after being cut. “These are novel living machines,” says Joshua Bongard, a computer scientist and robotics expert at the University of Vermont who co-led the new research. “They’re neither a traditional robot nor a known species of animal. It’s a new class of artifact: a living, programmable organism.” The new creatures were designed on a supercomputer at UVM—and then assembled and tested by biologists at Tufts University. “We can imagine many useful applications of these living robots that other machines can’t do,” says co-leader Michael Levin who directs the Center for Regenerative and Developmental Biology at Tufts, “like searching out nasty compounds or radioactive contamination, gathering microplastic in the oceans, traveling in arteries to scrape out plaque.” The results of the new research were published January 13 in the Proceedings of the National Academy of Sciences.

“As Xenobot “parents” move around their environment, they collect loose stem cells in their “mouths” that, over time, aggregate to create “offspring” Xenobots that develop to look just like their creators”
Bespoke living systems
People have been manipulating organisms for human benefit since at least the dawn of agriculture, genetic editing is becoming widespread, and a few artificial organisms have been manually assembled in the past few years—copying the body forms of known animals. But this research, for the first time ever, “designs completely biological machines from the ground up,” the team writes in their new study. With months of processing time on the Deep Green supercomputer cluster at UVM’s Vermont Advanced Computing Core, the team—including lead author and doctoral student Sam Kriegman—used an evolutionary algorithm to create thousands of candidate designs for the new life-forms. Attempting to achieve a task assigned by the scientists—like locomotion in one direction—the computer would, over and over, reassemble a few hundred simulated cells into myriad forms and body shapes. As the programs ran—driven by basic rules about the biophysics of what single frog skin and cardiac cells can do—the more successful simulated organisms were kept and refined, while failed designs were tossed out. After a hundred independent runs of the algorithm, the most promising designs were selected for testing.

“Designing and manufacturing reconfigurable organisms”
Then the team at Tufts, led by Levin and with key work by microsurgeon Douglas Blackiston, transferred the in silico designs into life. First they gathered stem cells, harvested from the embryos of African frogs, the species Xenopus laevis. (Hence the name “xenobots.”) These were separated into single cells and left to incubate. Then, using tiny forceps and an even tinier electrode, the cells were cut and joined under a microscope into a close approximation of the designs specified by the computer. Assembled into body forms never seen in nature, the cells began to work together. The skin cells formed a more passive architecture, while the once-random contractions of heart muscle cells were put to work creating ordered forward motion as guided by the computer’s design, and aided by spontaneous self-organizing patterns—allowing the robots to move on their own.
These reconfigurable organisms were shown to be able move in a coherent fashion—and explore their watery environment for days or weeks, powered by embryonic energy stores. Turned over, however, they failed, like beetles flipped on their backs. Later tests showed that groups of xenobots would move around in circles, pushing pellets into a central location—spontaneously and collectively. Others were built with a hole through the center to reduce drag. In simulated versions of these, the scientists were able to repurpose this hole as a pouch to successfully carry an object. “It’s a step toward using computer-designed organisms for intelligent drug delivery,” says Bongard, a professor in UVM’s Department of Computer Science and Complex Systems Center.

“Manufacturing reconfigurable organisms”
Living technologies
Many technologies are made of steel, concrete or plastic. That can make them strong or flexible. But they also can create ecological and human health problems, like the growing scourge of plastic pollution in the oceans and the toxicity of many synthetic materials and electronics. “The downside of living tissue is that it’s weak and it degrades,” say Bongard. “That’s why we use steel. But organisms have 4.5 billion years of practice at regenerating themselves and going on for decades.” And when they stop working—death—they usually fall apart harmlessly. “These xenobots are fully biodegradable,” say Bongard, “when they’re done with their job after seven days, they’re just dead skin cells.” Your laptop is a powerful technology. But try cutting it in half. Doesn’t work so well. In the new experiments, the scientists cut the xenobots and watched what happened. “We sliced the robot almost in half and it stitches itself back up and keeps going,” says Bongard. “And this is something you can’t do with typical machines.” Both Levin and Bongard say the potential of what they’ve been learning about how cells communicate and connect extends deep into both computational science and our understanding of life. “The big question in biology is to understand the algorithms that determine form and function,” says Levin. “The genome encodes proteins, but transformative applications await our discovery of how that hardware enables cells to cooperate toward making functional anatomies under very different conditions.”
To make an organism develop and function, there is a lot of information sharing and cooperation—organic computation—going on in and between cells all the time, not just within neurons. These emergent and geometric properties are shaped by bioelectric, biochemical, and biomechanical processes, “that run on DNA-specified hardware,” Levin says, “and these processes are reconfigurable, enabling novel living forms.” The scientists see the work presented in their new PNAS study—”A scalable pipeline for designing reconfigurable organisms,”—as one step in applying insights about this bioelectric code to both biology and computer science. “What actually determines the anatomy towards which cells cooperate?”
Levin asks. “You look at the cells we’ve been building our xenobots with, and, genomically, they’re frogs. It’s 100% frog DNA—but these are not frogs. Then you ask, well, what else are these cells capable of building? As we’ve shown, these frog cells can be coaxed to make interesting living forms that are completely different from what their default anatomy would be,” says Levin. He and the other scientists in the UVM and Tufts team—with support from DARPA’s Lifelong Learning Machines program and the National Science Foundation— believe that building the xenobots is a small step toward cracking what he calls the “morphogenetic code,” providing a deeper view of the overall way organisms are organized—and how they compute and store information based on their histories and environment.
Many people worry about the implications of rapid technological change and complex biological manipulations. “That fear is not unreasonable,” Levin says. “When we start to mess around with complex systems that we don’t understand, we’re going to get unintended consequences.” A lot of complex systems, like an ant colony, begin with a simple unit—an ant—from which it would be impossible to predict the shape of their colony or how they can build bridges over water with their interlinked bodies. “If humanity is going to survive into the future, we need to better understand how complex properties, somehow, emerge from simple rules,” says Levin.
Much of science is focused on “controlling the low-level rules. We also need to understand the high-level rules,” he says. “If you wanted an anthill with two chimneys instead of one, how do you modify the ants? We’d have no idea. I think it’s an absolute necessity for society going forward to get a better handle on systems where the outcome is very complex,” Levin says. “A first step towards doing that is to explore: how do living systems decide what an overall behavior should be and how do we manipulate the pieces to get the behaviors we want?” In other words, “this study is a direct contribution to getting a handle on what people are afraid of, which is unintended consequences,” Levin says—whether in the rapid arrival of self-driving cars, changing gene drives to wipe out whole lineages of viruses, or the many other complex and autonomous systems that will increasingly shape the human experience. “There’s all of this innate creativity in life,” says UVM’s Josh Bongard. “We want to understand that more deeply—and how we can direct and push it toward new forms.”
More information: Sam Kriegman el al., “A scalable pipeline for designing reconfigurable organisms,” PNAS (2019). www.pnas.org/cgi/doi/10.1073/pnas.1910837117
PREVIOUSLY
BIOHYBRID ROBOTS
https://spectrevision.net/2016/08/12/biohybrid-robots/
RECREATIONAL GENETICS
https://spectrevision.net/2015/05/08/recreational-genetics/
SWARMBOTS
https://spectrevision.net/2013/10/04/swarmbots/