16 YEAR OLD ASKS and ANSWERS
http://mnn.com/boy-discovers-microbe-that-eats-plastic
Boy discovers microbe that eats plastic / Jun 12 2009
“It’s not your average science fair when the 16-year-old winner manages to solve a global waste crisis. But such was the case at last May’s Canadian Science Fair in Waterloo, Ontario, where Daniel Burd, a high school student at Waterloo Collegiate Institute, presented his research on microorganisms that can rapidly biodegrade plastic.
NOTE: There are TWO high school students who discovered plastic-consuming microorganisms. The first was Daniel Burd. The second was Tseng I-Ching (last month), a high school student in Taiwan
Daniel had a thought it seems even the most esteemed PhDs hadn’t considered. Plastic, one of the most indestructible of manufactured materials, does in fact eventually decompose. It takes 1,000 years but decompose it does, which means there must be microorganisms out there to do the decomposing. Could those microorganisms be bred to do the job faster? That was Daniel’s question, and he put to the test with a very simple and clever process of immersing ground plastic in a yeast solution that encourages microbial growth, and then isolating the most productive organisms. The preliminary results were encouraging, so he kept at it, selecting out the most effective strains and interbreeding them. After several weeks of tweaking and optimizing temperatures Burd was achieved a 43 percent degradation of plastic in six weeks, an almost inconceivable accomplishment. With 500 billion plastic bags manufactured each year and a Pacific Ocean Garbage Patchthat grows more expansive by the day, a low-cost and nontoxic method for degrading plastic is the stuff of environmentalists’ dreams and, I would hazard a guess, a pretty good start-up company as well.
NOTE to the comment below: Yes there are certainly methods for decomposing plastic, but most are chemical in nature not organic, requiring high temperatures and chemical additives to cause the plasticizers to vaporize, for instance this patent on PVC extraction. There have been several successful bacteria-based solutions developed at theDepartment of Biotechnology in Tottori, Japan as well as the Department of Microbiology at the National University of Ireland, but both apply only to styrene compounds.
It goes without saying that these discoveries need to be tested to ensure, for instance, that the byproducts of organic decomposition are not carcinogenic (as in the case with mammalian metabolism of styrene and benzene). The processing of plastics by these methods would also have to be contained in highly controlled environments. So, no, we’re not talking about a magic panacea or a plastic-free paradise, but the innovative application of microorganisms to break down our most troublesome waste products is nevertheless a major scientific breakthrough. One of our readers pointed out a very interesting study in 2004 at the University of Wisconsin that isolated a fungus capable of biodegrading phenol-formaldehyde polymers previously thought to be non-biodegradable. Phenol polymers are produced at an annual rate of 2.2 million metric tons per year in the United States for many industrial and commercial applications including durable plastics.”
Comments : This story has generated a flurry of feedback since it was posted on June 12. Here’s a compilation of the best and brightest comments.”
FUNGUS (Pestalotiopsis Microspora)
http://fastcoexist.com/1679201/fungi-discovered-in-the-amazon-will-eat-your-plastic
Fungi Discovered In The Amazon Will Eat Your Plastic / by Michael Coren
“The Amazon is home to more species than almost anywhere else on earth. One of them, carried home recently by a group from Yale University, appears to be quite happy eating plastic in airless landfills. The group of students, part of Yale’s annual Rainforest Expedition and Laboratory with molecular biochemistry professor Scott Strobel, ventured to the jungles of Ecuador. The mission was to allow “students to experience the scientific inquiry process in a comprehensive and creative way.” The group searched for plants, and then cultured the microorganisms within the plant tissue. As it turns out, they brought back a fungus new to science with a voracious appetite for a global waste problem: polyurethane.

The common plastic is used for everything from garden hoses to shoes and truck seats. Once it gets into the trash stream, it persists for generations. Anyone alive today is assured that their old garden hoses and other polyurethane trash will still be here to greet his or her great, great grandchildren. Unless something eats it. The fungi, Pestalotiopsis microspora, is the first anyone has found to survive on a steady diet of polyurethane alone and–even more surprising–do this in an anaerobic (oxygen-free) environment that is close to the condition at the bottom of a landfill. Student Pria Anand recorded the microbe’s remarkable behavior and Jonathan Russell isolated the enzymes that allow the organism to degrade plastic as its food source. The Yale team published their findings in the journal Applied and Environmental Microbiology late last year concluding the microbe is “a promising source of biodiversity from which to screen for metabolic properties useful for bioremediation.” In the future, our trash compactors may simply be giant fields of voracious fungi. Who knows what the students in the rainforest will turn up next?”

photo: dhcp.tcgs.tc.edu.tw
STYROFOAM TOO?
http://mnn.com/high-school-girl-discovers-styrofoam-eating-bacterium
High school girl discovers Styrofoam-eating bacterium / Jun 13 2009
“I blogged about a Canadian student’s discovery of plastic-eating microorganisms last May. Just last month, another 16-year-old high school student (this time from Taiwan), Tseng I-Ching swept the world’s largest science fair in the Peoples Choice Category at the Intel International Science & Engineering Fair (ISEF) for her discovery of apolystyrene-decomposing bacterium derived from mealworm beetles. I-Ching vivisected more than 500 mealworm beetles to isolate the single bacterium that allows the mealworm to digest one of the most troublesome forms of waste on the planet — Styrofoam. For her discovery, I-Ching was awarded the top prize in the microbiology category along with four other prizes. The girl, nicknamed “Frog,” says her main career objective is to become a microbiologist and “save the world.” To that end, she spent the better part of her school year skipping classes to develop her innovative project isolating the “red bacteria” with the support of two leading microbiology scholars in Taipei. Her hard work got her in trouble at school (at one point she almost stopped her research project due to pressure from her school teachers) but she carried on and is now grateful she stuck with her passion. As she says, “I love to observe and find wonder from nature. I love to solve questions. This is how I started my project.” There have been two successful bacteria-based solutions for styrene decomposition developed at the Department of Biotechnology in Tottori, Japan, as well as the Department of Microbiology at the National University of Ireland. Both rely upon a patented soil organism called Pseudomonas putida.
Polystyrene is the bad boy of the petrochemical industry. In addition to the highly toxic chemicals required to manufacture polysterene products (namely benzene), expanded polystyrene foam requires ozone-depleting HCFC’s (CFC’s used to be used to make Styrofoam, but they have been banned for the most part). Then once disposed, it basically NEVER decomposes. It does however break apart into smaller granules, but because of its light weight, those particles quickly become both airborne and waterborne, where they wreck havoc on the ocean food chain. The U.S. disposes of about 25 billion Styrofoam cups every year and tons more extruded and expanded polystyrene packaging material. It’s a big, big problem. Biodegradable alternatives are now hitting the market, but hopefully Tseng I-Ching’s small discovery will help give to give existing Styrofoam waste a proper burial.”
GARBAGE as LANDMASS
http://digitaluniverse.net/upcycling/articles/view/135971/
The World’s Largest “Landfill” is in the Middle of the Ocean
BY David Sokoll / February 23, 2009
“There is a large part of the central Pacific Ocean that no one ever visits and only a few ever pass through. Sailors avoid it like the plague for it lacks the wind they need to sail. Fisherman leave it alone because its lack of nutrients makes it an oceanic desert. This area includes the “horse latitudes,” where stock transporters in the age of sail got stuck, ran out of food and water and had to jettison their horses and other livestock. Surprisingly, this is the largest ocean realm on our planet, being about the size of Africa – over ten million square miles. A huge mountain of air, which has been heated at the equator, and then begins descending in a gentle clockwise rotation as it approaches the North Pole, creates this ocean realm. The circular winds produce circular ocean currents which spiral into a center where there is a slight down-welling. Scientists know this atmospheric phenomenon as the subtropical high, and the ocean current it creates as the north Pacific central or sub-tropical gyre. Because of the stability of this gentle maelstrom, the largest uniform climatic feature on earth is also an accumulator of the debris of civilization. Anything that floats, no matter where it comes from on the north Pacific Rim or ocean, ends up here, sometimes after drifting around the periphery for twelve years or more. Historically, this debris did not accumulate because it was eventually broken down by microorganisms into carbon dioxide and water. Now, however, in our battle to store goods against natural deterioration, we have created a class of products that defeats even the most creative and insidious bacteria. They are plastics.
Plastics are now virtually everywhere in our modern society. We drink out of them, eat off of them, sit on them, and even drive in them. They’re durable, lightweight, cheap, and can be made into virtually anything. But it is these useful properties of plastics, which make them so harmful when they end up in the environment. Plastics, like diamonds, are forever! If plastic doesn’t biodegrade, what does it do? It “photo-degrades” – a process in which it is broken down by sunlight into smaller and smaller pieces, all of which are still plastic polymers, eventually becoming individual molecules of plastic, still too tough for anything to digest. For the last fifty-odd years, every piece of plastic that has made it from our shores to the Pacific Ocean, has been breaking down and accumulating in the central Pacific gyre. Oceanographers like Curtis Ebbesmeyer, the world’s leading flotsam expert, refer to it as the great Pacific Garbage Patch. The problem is that it is not a patch, it’s the size of a continent, and it’s filling up with floating plastic waste. My research has documented six pounds of plastic for every pound of plankton in this area. My latest 3-month round trip research voyage just completed in Santa Barbara this week, (our departure was covered by SBNP) got closer to the center of the Garbage Patch than before and found levels of plastic fragments that were far higher for hundreds of miles. We spent weeks documenting the effects of what amounts to floating plastic sand of all sizes on the creatures that inhabit this area. Our photographers captured images of jellyfish hopelessly entangled in frayed line, and transparent filter feeding organisms with colorful plastic fragments in their bellies.
As we drifted in the center of this system, doing underwater photography day and night, we began to realize what was happening. A paper plate thrown overboard just stayed with us, there was no wind or current to move it away. This is where all those things that wash down rivers to the sea end up. On October 10, during our return trip to Santa Barbara, we discovered something never before documented-a Langmuir Windrow of plastic debris. Circular ocean currents with contrary rotation create long lines of material, visible from above as streaks on the ocean. Normally these are formed by planktonic organisms or foam, but we discovered one made of plastic. Everything from huge hawsers to tiny fragments were formed into a miles long line. We picked up hundreds of pounds of netting of all types bailed together in this system along with every type and size of debris imaginable. Sometimes, windrows like this drift down over the Hawaiian Islands. That is when Waimanalo Beach on Oahu gets coated with blue green plastic sand, along with staggering amounts of larger debris. Farther to the northwest, at the Northwestern Hawaiian Islands Coral Reef Ecosystem Reserve, monk seals, the most endangered mammal species in the United States, get entangled in debris, especially cheap plastic nets lost or discarded by the fishing industry. Ninety percent of Hawaiian green sea turtles nest here and eat the debris, mistaking it for their natural food, as do Laysan and Black Footed Albatross. Indeed, the stomach contents of Laysan Albatross look like the cigarette lighter shelf at a convenience store they contain so many of them.

It’s not just entanglement and indigestion that are problems caused by plastic debris, however. There is a darker side to pollution of the ocean by ubiquitous plastic fragments. As these fragments float around , they accumulate the poisons we manufacture for various purposes that are not water-soluble. It turns out that plastic polymers are sponges for DDT, PCBs and nonylphenols -oily toxics that don’t dissolve in seawater. Plastic pellets have been found to accumulate up to one million times the level of these poisons that are floating in the water itself. These are not like heavy metal poisons which affect the animal that ingests them directly. Rather, they are what might be called “second generation “ toxics. Animals have evolved receptors for elaborate organic molecules called hormones, which regulate brain activity and reproduction. Hormone receptors cannot distinguish these toxics from the natural estrogenic hormone, estradiol, and when the pollutants dock at these receptors instead of the natural hormone, they have been shown to have a number of negative effects in everything from birds and fish to humans. The whole issue of hormone disruption is becoming one of, if not the biggest environmental issue of the 21st Century. Hormone disruption has been implicated in lower sperm counts and higher ratios of females to males in both humans and animals. Unchecked, this trend is a dead end for any species.
A trillion trillion vectors for our worst pollutants are being ingested by the most efficient natural vacuum cleaners nature ever invented, the mucus web feeding jellies and salps (chordate jellies that are the fastest growing multicellular organisms on the planet) out in the middle of the ocean. These organisms are in turn eaten by fish and then, certainly in many cases, by humans. We can grow pesticide free organic produce, but can nature still produce a pesticide free organic fish? After what I have witnessed first hand in the Pacific, I have my doubts. I am often asked why we can’t vacuum up the particles. In fact, it would be more difficult than vacuuming up every square inch of the entire United States, it’s larger and the fragments are mixed below the surface down to at least 30 meters. Also, untold numbers of organisms would be destroyed in the process. Besides, there is no economic resource that would be directly benefited by this process. We have not yet learned how to factor the health of the environment into our economic paradigm. We need to get to work on this calculus quickly, for a stock market crash will pale by comparison to an ecological crash on an oceanic scale. I know that when people think of the deep blue ocean, they see images of pure, clean, unpolluted water. After we sample the surface water in the central Pacific, I often dive over with a snorkel and a small aquarium net. I have yet to come back after a fifteen minute swim without plastic fragments for my collection. I can no longer see pristine images when I think of the briny deep. Neither can I imagine any “beach cleanup” type of solution. Only elimination of the source of the problem can result in an ocean nearly free from plastic, and the desired result will only be seen by citizens of the third millennium AD. The battle to change the way we produce and consume plastics has just begun, but I believe it is essential that it be fought now. The levels of plastic particulates in the Pacific have at least tripled in the last ten years and a tenfold increase in the next decade is not unreasonable. Then, sixty times more plastic than plankton will float on its surface.”

UNDER ESTIMATED
http://californiawatch.org/dailyreport/plastic-pollution-ocean-likely-underestimated
Plastic pollution in ocean likely underestimated, researchers say
by Susanne Rust / April 30, 2012
“The cause célèbre of plastic litter in the ocean is the Texas-sized, swirling island of plastic debris thousands of miles off the coast of California in the Pacific Ocean. But researchers from the Universities of Washington and Delaware and the Sea Education Association in Woods Hole, Mass., say the story is much bigger, and scarier, than that. They say scientists have only skimmed the surface on the devastating pollution caused by plastic debris in the ocean, and the research community is likely underestimating the amount of plastic in the ocean. According to new research, natural ocean processes such as wind, drag, turbulence and wave height can push the plastic deep down, where it floats along, suspended underwater and unobserved by people examining the ocean’s surface. The research is published in the journal Geophysical Research Letters.
Giora Proskurowski, the University of Washington researcher, said his eureka moment happened while working on a research sailboat 2,000 miles off the West Coast. Looking overboard, as the wind died down, he saw little white particles littered over the surface. “It was like a photograph coming into focus,” he said. As the rippled waves on the surface died, the white specks appeared. But half an hour later, as the wind picked it up, they all disappeared. He decided to take samples from below the surface, at a depth of 16 feet. And there, he discovered plastic moving through the water column, even though he couldn’t see it on the surface. Taking the experiment a step further, Proskurowski looked at data that had been collected by the Sea Education Association from the 1990s and early 2000s. He also collected more samples from the North Atlantic at different depths, going down as far as 100 feet. “Almost every tow we did contained plastic, regardless of the depth,” he said. Using that data with wind measurements that had been collected, Proskurowski and his team were able to devise a simple model that can potentially be used by others, including non-scientists, to match wind data with surface plastic collections to estimate how much was missed by only skimming the surface. “In order to make an accurate estimate, you need to consider the wind conditions under which that sample was taken,” he said. The team is hoping others will use this model – which is available upon request – to evaluate current investigations of plastic pollution in the ocean.
“I think what this research really shows is that I can go out in the middle of the ocean – five days away from land – into one of the remotest parts of the planet, and scoop up plastic,” he said. “I think it’s a stark reminder of the impact our society has on the planet.” “And it should make us realize that any systematic cleanup approach we might make is going to be difficult,” he said. The American Chemistry Council, the chemical and plastics industry trade group, could not be reached for comment. Capt. Charles Moore of the Algalita Marine Research Foundation in Long Beach said the research supports the work his own organization has done, which also has found that plastic sinks in the water column and its presence is underestimated. “I’d suggest they start analyzing the sediments of the mouths of urban rivers,” where plastics heavier than water get mixed in with soils, Moore said. Moore said the presence of plastic in the ocean is concerning for a variety of reasons. His foundation has discovered that reef-like structures are forming on the plastic, with crabs, sea anemones, barnacles and even coral heads living on the plastic jetsam floating in the ocean. These reefs alter the deep ocean by blocking sunlight and alter the ocean’s chemistry. He said the plastics also are toxic, and as animals feed on them, these pollutants enter the food chain, carrying and amplifying them as animal after animal is exposed. “Then there’s just the straight ingestion issue,” he said, with evidence that whales, turtles and fish are filled with these plastics. “I wish there were a simple answer to fixing this,” he said. “But our economy is based on throwaway, packaged items.”
UPCYCLING
http://digitaluniverse.net/upcycling/
http://wiserearth.org/group/BerkanaExchange
http://newscientist.com/article/dn14764
Plastic-munching bugs turn waste bottles into cash
BY Colin Barras / 19 September 2008
“Newly discovered bacterial alchemists could help save billions of plastic bottles from landfill. The Pseudomonas strains can convert the low-grade PET plastic used in drinks bottles into a more valuable and biodegradable plastic called PHA. PHA is already used in medical applications, from artery-supporting tubes called stents to wound dressings. The plastic can be processed to have a range of physical properties. However, one of the barriers to PHA reaching wider use is the absence of a way to make it in large quantities. The new bacteria-driven process – termed upcycling – could address that, and make recycling PET bottles more economically attractive.
PET bugs
Although billions of plastic bottles are made each year, few are ultimately recycled. Just 23.5% of US bottles were recycled in 2006. This is because the recycling process simply converts the low value PET bottles into more PET, says Kevin O’Connor at University College Dublin, Ireland. “We wanted to see if we could turn the plastic into something of higher value in an environmentally friendly way,” he says. O’Connor and colleagues knew that heating PET in the absence of oxygen – a process called pyrolysis – breaks it down into terephthalic acid (TA) and a small amount of oil and gas. They also knew that some bacteria can grow and thrive on TA, and that other bacteria produce a high-value plastic PHA when stressed. So they wondered whether any bacteria could both feed on TA and convert it into PHA.
Bacteria hunt
“It was a long shot to be honest,” says O’Connor. His team studied cultures from around the world known to grow on TA, but none produced PHA. So they decided to look for undiscovered strains, in environments that naturally contain TA. Analysing soil bacteria from a PET bottle processing plant, which are likely to be exposed to small quantities of TA, yielded 32 colonies that could survive in the lab using TA as their only energy source. After 48 hours they screened each culture for PHA. Three cultures, all similar to known strains of Pseudomonas, accumulated detectable quantities of the valuable plastic. The next step is to improve the efficiency of the process, says O’Connor. “A quarter to a third of each cell is filled with plastic – we want to increase that to 50 to 60%.”
Less landfill
Sudesh Kumar, a microbiologist at the University of Science, Malaysia, in Penang, is impressed with the study. “There are many other systems that are economically more viable to produce PHA with better material properties,” he says. “But Kevin’s work offers an interesting novel approach to solve the problem of PET accumulation in landfill dumps.” But it is still unlikely that using the new approach alone will appeal to industry, O’Connor says. “Working with this kind of environmental technology in isolation, the chances of success are reduced,” he says. The best approach, he continues, would be to use the new bacteria as just one part of a bio-refinery capable of upcycling an array of waste products in an environmentally friendly way. {Journal reference: Environmental Science and Technology (DOI: 10.1021/es801010e)}”
CONTACT
Kevin O’Connor
http://ucd.ie/research/success/featuredacademics/drkevinoconnor/
http://ucd.ie/research/people/biomolecularbiomedscience/drkevinedwardoconnor/home/
email : kevin.oconnor [at] ucd [dot] ie
ORV ALGUITA
http://alguita.com/orv_alguita.html
http://alguita.com/research_papers.html
http://algalita.org/research.html
http://algalita.org/Maps_Home.html
DOWN the RABBITHOLE
http://en.wikipedia.org/wiki/Polyethylene_terephthalate
http://en.wikipedia.org/wiki/Polyhydroxyalkanoates
http://en.wikipedia.org/wiki/Pseudomonas
http://en.wikipedia.org/wiki/Sphingomonas
HEY THANKS! CANADIAN TEEN DECOMPOSES PLASTIC BAG in THREE MONTHS
http://blog.wired.com/wiredscience/2008/05/teen-decomposes.html
by Brandon Keim / May 23, 2008
“The Waterloo, Ontario high school junior figured that something must make plastic degrade, even if it does take millennia, and that something was probably bacteria. (Hey, at between one-half and 90 percent of Earth’s biomass, bacteria’s a pretty safe bet for any biological mystery.) The Record reports that Burd mixed landfill dirt with yeast and tap water, then added ground plastic and let it stew. The plastic indeed decomposed more quickly than it would in nature; after experimenting with different temperatures and configurations, Burd isolated the microbial munchers. One came from the bacterial genus Pseudomonas, and the other from the genus Sphingomonas. Burd says this should be easy on an industrial scale: all that’s needed is a fermenter, a growth medium and plastic, and the bacteria themselves provide most of the energy by producing heat as they eat. The only waste is water and a bit of carbon dioxide. Amazing stuff. I’ll try to get an interview with this young man who may have managed to solve one of the most intractable environmental dilemmas of our time. And I can’t help but wonder whether his high school already had its prom. If he doesn’t get to be king, there’s no justice in this world.”
AGREED: DANIEL BURD for PROM KING
http://apps.ysf-fsj.ca/virtualcwsf/projectdetails.php?id=1390
“My name is Daniel Burd, a grade 11 student at Waterloo Collegiate Institute. I performed my first “science” experiment involving planting and observing growth of different types of tomato seeds on the balcony of our apartment in Waterloo eleven years ago. Since that time, the ideas and concepts behind the way things work have constantly aroused my interest and have posed numerous questions for me to consider. At school, I am on ABCD Student Council, Charity Controller, Environment club, a peer tutor, and Norse Star newspaper. When I was five years old, I started to play the piano and I have completed my grade 8 piano and grade 2 rudiments at the RCM. Currently, I am learning improvisation and jazz. My jazz music role model is Oscar Peterson. I am a member of Nordic Skiing club, ROW swimming club and Waterloo Tennis Club where I am training for tournaments. I am a volunteer at K-W Science and Technology Children’s Museum. I help organize heritage events in K-W area and I run a charity dog-walking business in my neighborhood for people with disabilities. I fluently speak English, French and Russian and I enjoy spending free time with my friends.”
NEVER MIND SCHOLARSHIPS, GET HIM AN INVESTOR
– The Manning Innovation Achievement Award – $500.00
– Dalhousie University Faculty of Science Entrance Scholarship
Senior Gold Medallist – $4000 Entrance Scholarship
– NSERC Undergraduate Student Research Award
Senior Gold Medallist – $5 625.00
– UBC Science (Vancouver) Entrance Award
Senior Gold Medallist – $4000 Entrance Scholarship
– University of Ottawa Entrance Scholarship
Senior Gold Medallist – $20,000 Entrance Scholarship
($5,000 each year for 4
years)
– Senior Silver Medallist – $3000 Entrance Scholarship
– The University of Western Ontario Scholarship
Gold Medallist – $2000 Entrance Scholarship
– The University of Western Ontario Scholarship
Silver Medallist – $1500 Entrance Scholarship
– Silver Medal – Environmental Innovation – $700.00
– Gold Medal – Biotechnology & Pharmaceutical Sciences – $1 500.00
– EnCana Platinum Award – Best Senior Project – $5 000.00
– EnCana Best in Fair Award – $10 000.00
Total $57 825.00
YEAST, TAP WATER, DIRT
http://news.therecord.com/article/354044
WCI student isolates microbe that lunches on plastic bags
BY Karen Kawawada
“Getting ordinary plastic bags to rot away like banana peels would be an environmental dream come true. After all, we produce 500 billion a year worldwide and they take up to 1,000 years to decompose. They take up space in landfills, litter our streets and parks, pollute the oceans and kill the animals that eat them. Now a Waterloo teenager has found a way to make plastic bags degrade faster — in three months, he figures.
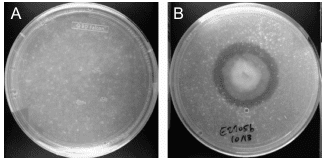
Daniel Burd’s project won the top prize at the Canada-Wide Science Fair in Ottawa. He came back with a long list of awards, including a $10,000 prize, a $20,000 scholarship, and recognition that he has found a practical way to help the environment. Daniel, a 16-year-old Grade 11 student at Waterloo Collegiate Institute, got the idea for his project from everyday life. “Almost every week I have to do chores and when I open the closet door, I have this avalanche of plastic bags falling on top of me,” he said. “One day, I got tired of it and I wanted to know what other people are doing with these plastic bags.” The answer: not much. So he decided to do something himself. He knew plastic does eventually degrade, and figured microorganisms must be behind it. His goal was to isolate the microorganisms that can break down plastic — not an easy task because they don’t exist in high numbers in nature.
First, he ground plastic bags into a powder. Next, he used ordinary household chemicals, yeast and tap water to create a solution that would encourage microbe growth. To that, he added the plastic powder and dirt. Then the solution sat in a shaker at 30 degrees. After three months of upping the concentration of plastic-eating microbes, Burd filtered out the remaining plastic powder and put his bacterial culture into three flasks with strips of plastic cut from grocery bags. As a control, he also added plastic to flasks containing boiled and therefore dead bacterial culture. Six weeks later, he weighed the strips of plastic. The control strips were the same. But the ones that had been in the live bacterial culture weighed an average of 17 per cent less. That wasn’t good enough for Burd. To identify the bacteria in his culture, he let them grow on agar plates and found he had four types of microbes. He tested those on more plastic strips and found only the second was capable of significant plastic degradation.
Next, Burd tried mixing his most effective strain with the others. He found strains one and two together produced a 32 per cent weight loss in his plastic strips. His theory is strain one helps strain two reproduce. Tests to identify the strains found strain two was Sphingomonas bacteria and the helper was Pseudomonas. A researcher in Ireland has found Pseudomonas is capable of degrading polystyrene, but as far as Burd and his teacher Mark Menhennet know — and they’ve looked — Burd’s research on polyethelene plastic bags is a first. Next, Burd tested his strains’ effectiveness at different temperatures, concentrations and with the addition of sodium acetate as a ready source of carbon to help bacteria grow. At 37 degrees and optimal bacterial concentration, with a bit of sodium acetate thrown in, Burd achieved 43 per cent degradation within six weeks.
The plastic he fished out then was visibly clearer and more brittle, and Burd guesses after six more weeks, it would be gone. He hasn’t tried that yet. To see if his process would work on a larger scale, he tried it with five or six whole bags in a bucket with the bacterial culture. That worked too. Industrial application should be easy, said Burd. “All you need is a fermenter . . . your growth medium, your microbes and your plastic bags.” The inputs are cheap, maintaining the required temperature takes little energy because microbes produce heat as they work, and the only outputs are water and tiny levels of carbon dioxide — each microbe produces only 0.01 per cent of its own infinitesimal weight in carbon dioxide, said Burd. “This is a huge, huge step forward . . . We’re using nature to solve a man-made problem.” Burd would like to take his project further and see it be used. He plans to study science at university, but in the meantime he’s busy with things such as student council, sports and music. “Dan is definitely a talented student all around and is poised to be a leading scientist in our community,” said Menhennet, who led the school’s science fair team but says he only helped Burd with paperwork.”
STYROFOAM
http://portal.acs.org/portal/acs/corg/content?_nfpb=true&_pageLabel=PP_ARTICLEMAIN&node_id=222&content_id=CTP_003309&use_sec=true&sec_url_var=region1
Microbes convert ‘Styrofoam™’ into biodegradable plastic / 02/23/2006
“Bacteria could help transform a key component of disposable cups, plates and utensils into a useful eco-friendly plastic, significantly reducing the environmental impact of this ubiquitous, but difficult-to- recycle waste stream, according to a study scheduled to appear in the April 1 issue of the American Chemical Society journal, Environmental Science & Technology. The microbes, a special strain of the soil bacterium Pseudomonas putida, converted polystyrene foam — commonly known as Styrofoam™ — into a biodegradable plastic, according to Kevin O’Connor, Ph.D., of University College Dublin, the study’s corresponding author. The study is among the first to investigate the possibility of converting a petroleum-based plastic waste into a reusable biodegradable form. O’Connor and his colleagues from Ireland and Germany, utilized pyrolysis, a process that transforms materials by heating them in the absence of oxygen, to convert polystyrene — the key component of many disposable products — into styrene oil. The researchers then supplied this oil to P. putida, a bacterium that can feed on styrene, which converted the oil into a biodegradable plastic known as PHA (polyhydroxyalkanoates). The process might also be used to convert other types of discarded plastics into PHA, according to O’Connor. PHA has numerous uses in medicine and can be used to make plastic kitchenware, packaging film and other disposable items. The biodegradable plastic is resistant to hot liquids, greases and oils, and can have a long shelf life. But unlike polystyrene, it readily breaks down in soil, water, septic systems and backyard composts. Worldwide, more than 14 million metric tons of polystyrene are produced annually, according to the U.S. Environmental Protection Agency. Most of this ends up in landfills. Although polystyrene represents less than 1 percent of solid waste generated in the United States, at least 2.3 million tons of it is dumped in U.S. landfills each year. Only 1 percent of polystyrene waste is currently recycled, the authors note.”
SEE ALSO
Anthony J. Sinskey, Sc.D.
http://web.mit.edu/biology/www/facultyareas/facresearch/sinskey.html
http://hst.mit.edu/public/people/faculty/facultyBiosketch.jsp?key=Sinskey
e-mail : asinskey [at] mit [dot] edu
Oliver Peoples, Ph.D. / Founder / Chief Scientific Officer, Metabolix
http://metabolix.com/
http://metabolix.com/natures%20plastic/coretechnology.html
http://metabolix.com/biotechnology%20foundation/biotechnologyfoundation.html
http://metabolix.com/sustainable%20solutions/sustainablesolutions.html
http://metabolix.com/resources/researchlinks.html
e-mail : peoples [at] metabolix [dot] com
BIOPLAST
http://bioplast.com.tr/bioplast.html
http://bioplast.com.tr/bio-bozunur-plastics.html
PSEUDOMONAS GENOME DATABASE
http://pseudomonas.com/
ALSO CAUSES SNOWFLAKES
http://efluxmedia.com/Bacteria_the_Main_Ingredient_in_Snowflakes_Scientists_Say
Bacteria – The Main Ingredient in Snowflakes, Scientists Say
BY Max Brenn / February 29th 2008
“One might rethink playing with snow or walking in the rain as a new study by scientists from the Louisiana State University revealed that snow and rain might form mostly on bacteria in the clouds. Scientists have long known that the ice crystals in clouds, which become rain or snow, need to cling to some kind of particle, called ice nucleators, in order to form in temperatures above minus 40 degrees Celsius. Microbiologist Brent Christner at Louisiana State University sampled snow from Antarctica, France, and the Yukon and found that as much as 85 percent of the nuclei were bacteria, he said in a telephone interview with the Associated Press. “Every snow and ice sample we’ve looked at, we found biological ice nucleators. Here’s a component that has been completely ignored to date,” Christner said. The most common bacteria found was Psedomonas syringae, which can cause disease in several types of plants (tomatoes, green beams and other similar plants). The bacterium was found in 20 samples of snow from around the world and subsequent research has also found it in summer rainfall in Louisiana. Scientists have sought ways to eliminate this bacterium in time. Now they wonder whether this elimination would result in less rain or snow, or would soot and dust be the major generators of precipitation. “The question is, are they a good guy or a bad guy. And I don’t have the answer to that,” Christner said, quoted by the same source. One thing is for sure. Bacteria that infect plants may multiply on the plants’ leaves and drift into the atmosphere. These bacteria could then cause precipitation and land on another plant, where the life cycle could continue, Christner said. Virginia K.Walker, a biologist at Queen’s University in Kingston, Ontario, Canada said other studies have found bacteria serving as snow nuclei, but this is the first to identify it as Pseudomonas. “It’s one of those great bacteria…you can find them anywhere. They are really interesting,” Walker said. The study, supported by a Louisiana State University research grant and by the National Science Foundation and the Earth Institute at Columbia University, was published in today’s edition of the journal Science.”
CONTACT
Brent C. Christner
http://biology.lsu.edu/faculty_listings/fac_pages/bchristner.html
http://brent.xner.net/
email : xner [at] lsu [dot] edu
ABSTRACT
http://biology.lsu.edu/highlights/christner.html
Ubiquity of Biological Ice Nucleators in Snowfall
“Despite the integral role of ice nucleators (IN) in atmospheric processes leading to precipitation, their sources and distributions have not been well established. We examined IN in snowfall from mid- and high-latitude locations and found that the most active were biological in origin. Of the IN larger than 0.2 micrometer that were active at temperatures warmer than -7°C, 69 to 100% were biological, and a substantial fraction were bacteria. Our results indicate that the biosphere is a source of highly active IN and suggest that these biological particles may affect the precipitation cycle and/or their own precipitation during atmospheric transport”
ONE BILLION per QUART
http://time.com/time/magazine/article/0,9171,894282,00.html
Bugs in the Reactor / Oct. 05, 1959
“Los Alamos’ Omega West is a swimming-pool-type research reactor whose fuel rods are suspended under 25 ft. of water, which acts not only as coolant and moderator but also shields its human operators from radioactivity. In the spring of 1958, physicists peering down through it saw that the water was getting cloudy. They called Chemist-Bacteriologist Eric B. Fowler of the laboratory’s radioactive-waste disposal group, who found that it was swarming with microorganisms, about i billion per quart. The bugs turned out to be rod-shaped bacteria of the genus Pseudomonas, which were feeding on resin and felt in the water purifying system. The fierce radiation in the reactor appeared to bother the bacteria hardly at all. When the reactor was shut down but still highly radioactive, they multiplied fast. Even when it was running full blast, they held their own. Since they normally divide every 20 minutes or so, this meant that radiation was killing only about as many as managed to live and divide. Just how much radiation the Pseudomonas got is hard to estimate, because the water circulates at varying distances from the core of the reactor, but Dr. Fowler thinks they may have absorbed more than 10 million rep (roentgen equivalent physical) in an eight-hour day, which is 10,000 times the dose that is fatal to man. Many other microorganisms must have got into Omega West’s deadly water; only the Pseudomonas survived. Perhaps the Pseudomonas have natural resistance to radiation. More likely, under the bombardment of Omega’s radiation, normal Pseudomonas underwent mutation, producing a special strain capable of surviving in this atomic blast. This ability to transform themselves quickly to cope with new conditions is a specialty of humble bacteria, whose constitutions are relatively simple. It is an ability that higher animals cannot emulate, but may have reason to envy.”
PSEUDOMONAS and YOU
http://horizonpress.com/pseudo
http://horizonpress.com/gateway/pseudomonas
“The Taxonomy of Pseudomonas
The studies on the taxonomy of this complicated genus groped their way in the dark while following the classical procedures developed for the description and identification of the organisms involved in sanitary bacteriology during the first decades of the twentieth century. This situation sharply changed with the proposal to introduce as the central criterion the similarities in the composition and sequences of macromolecules components of the ribosomal RNA. The new methodology clearly showed that the genus Pseudomonas, as classical defined, consisted in fact of a conglomerate of genera that could clearly be separated into five so-called rRNA homology groups. Moreover, the taxonomic studies suggested an approach that might proved useful in taxonomic studies of all other prokaryotic groups. A few decades after the proposal of the new genus Pseudomonas by Migula in 1894, the accumulation of species names assigned to the genus reached alarming proportions. At the present moment, the number of species in the current list has contracted more than ten-fold. In fact, this approximated reduction may be even more dramatic if one considers that the present list contains many new names, i.e., relatively few names of the original list survived in the process. The new methodology and the inclusion of approaches based on the studies of conservative macromolecules other than rRNA components, constitutes an effective prescription that helped to reduce Pseudomonas nomenclatural hypertrophy to a manageable size.
Genome Diversity of Pseudomonas aeruginosa
The G+C rich Pseudomonas aeruginosa chromosome consists of a conserved core and a variable accessory part. The core genomes of P. aeruginosa strains are largely collinear, exhibit a low rate of sequence polymorphism and contain few loci of high sequence diversity, notably the pyoverdine locus, the flagellar regulon, pilA and the O-antigen biosynthesis locus. Variable segments are scattered throughout the genome of which about one third are immediately adjacent to tRNA or tmRNA genes. The three known hot spots of genomic diversity are caused by the integration of genomic islands of the pKLC102 / PAGI-2 family into tRNALys or tRNAGly genes. The individual islands differ in their repertoire of metabolic genes, but share a set of syntenic genes that confer their horizontal spread to other clones and species. Colonization of atypical disease habitats predisposes to deletions, genome rearrangements and accumulation of loss-of-function mutations in the P. aeruginosa chromosome. The P. aeruginosa population is characterized by a few dominant clones widespread in disease and environmental habitats. The genome is made up of clone-typical segments in core and accessory genome and of blocks in the core genome with unrestricted gene flow in the population.
Oligonucleotide Usage Signatures of the Pseudomonas putida KT2440 Genome
Di- to pentanucleotide usage and the list of the most abundant octa- to tetradecanucleotides are useful measures of the bacterial genomic signature. The Pseudomonas putida KT2440 chromosome is characterized by strand symmetry and intra-strand parity of complementary oligonucleotides. Each tetranucleotide occurs with similar frequency on the two strands. Tetranucleotide usage is biased by G+C content and physicochemical constraints such as base stacking energy, dinucleotide propeller twist angle or trinucleotide bendability. The 105 regions with atypical oligonucleotide composition can be differentiated by their patterns of oligonucleotide usage into categories of horizontally acquired gene islands, multidomain genes or ancient regions such as genes for ribosomal proteins and RNAs. A species-specific extragenic palindromic sequence is the most common repeat in the genome that can be exploited for the typing of P. putida strains. In the coding sequence of P. putida LLL is the most abundant tripeptide.
Genetic Tools for Pseudomonas
Genetic tools are required to take full advantage of the wealth of information generated by genome sequencing efforts, and ensuing global gene and protein expression analyses. Although the development of genetic tools has generally not kept up with the sequencing pace, substantial progress has been made in this arena. PCR- and recombination-based strategies allowed construction of whole genome expression and transposon insertion libraries. Similar strategies combined with improved transformation protocols facilitate high- throughput construction of deletion alleles and development of a broad-host-range mini-Tn7 chromosome integration system. While to date most of these tools and methods have been developed for and applied in P. aeruginosa, they will most likely also be applicable to other Pseudomonas with appropriate modifications.
Molecular Biology of Cell-Surface
Polysaccharides in Pseudomonas aeruginosa: From Gene to Protein Function
Cell-surface polysaccharides play diverse roles in the bacterial “lifestyle”. They serve as a barrier between the cell wall and the environment, mediate host-pathogen interactions, and form structural components of biofilms. These polysaccharides are synthesized from nucleotide-activated precursors and, in most cases, all the enzymes necessary for biosynthesis, assembly and transport of the completed polymer are encoded by genes organized in dedicated clusters within the genome of the organism. Lipopolysaccharide is one of the most important cell-surface polysaccharides, as it plays a key structural role in outer membrane integrity, as well as being an important mediator of host-pathogen interactions. The genetics for the biosynthesis of the so-called A-band (homopolymeric) and B-band (heteropolymeric) O antigens have been clearly defined, and a lot of progress has been made toward understanding the biochemical pathways of their biosynthesis. The exopolysaccharide alginate is a linear copolymer of ß-1,4-linked D-mannuronic acid and L-guluronic acid residues, and is responsible for the mucoid phenotype of late-stage cystic fibrosis disease. The pel and psl loci are two recently discovered gene clusters that also encode exopolysaccharides found to be important for biofilm formation. Rhamnolipid is a biosurfactant whose production is tightly regulated at the transcriptional level, but the precise role that it plays in disease is not well understood at present. Protein glycosylation, particularly of pilin and flagellin, is a recent focus of research by several groups and it has been shown to be important for adhesion and invasion during bacterial infection. Pseudomonas aeruginosa Virulence and Pathogenesis Issues Regulation of gene expression can occur through cell-cell communication or quorum sensing (QS) via the production of small molecules called autoinducers. QS is known to control expression of a number of virulence factors. Another form of gene regulation which allows the bacteria to rapidly adapt to surrounding changes is through environmental signaling. Recent studies have discovered that anaerobiosis can significantly impact the major regulatory circuit of QS. This important link between QS and anaerobiosis has a significant impact on production of virulence factors of this organism.
Pseudomonas aeruginosa
Biofilms: Impact of Small Colony Variants on Chronic Persistent Infections
The achievements of medical care in industrialised societies are markedly impaired due to chronic opportunistic infections that have become increasingly apparent in immunocompromised patients and the ageing population. Chronic infections remain a major challenge for the medical profession and are of great economic relevance because traditional antibiotic therapy is usually not sufficient to eradicate these infections. One major reason for persistence seems to be the capability of the bacteria to grow within biofilms that protects them from adverse environmental factors. Pseudomonas aeruginosa is not only an important opportunistic pathogen and causative agent of emerging nosocomial infections but can also be considered a model organism for the study of diverse bacterial mechanisms that contribute to bacterial persistence. In this context the elucidation of the molecular mechanisms responsible for the switch from planctonic growth to a biofilm phenotype and the role of inter-bacterial communication in persistent disease should provide new insights in P. aeruginosa pathogenicity, contribute to a better clinical management of chronically infected patients and should lead to the identification of new drug targets for the development of alternative anti-infective treatment strategies.
Antibiotic Resistance in Pseudomonas
Pseudomonas aeruginosa is a highly relevant opportunistic pathogen. One of the most worrisome characteristics of P. aeruginosa consists in its low antibiotic susceptibility. This low susceptibility is attributable to a concerted action of multidrug efflux pumps with chromosomally-encoded antibiotic resistance genes and the low permeability of the bacterial cellular envelopes. Besides intrinsic resistance, P. aeruginosa easily develop acquired resistance either by mutation in chromosomally-encoded genes, either by the horizontal gene transfer of antibiotic resistance determinants. Development of multidrug resistance by P. aeruginosa isolates requires several different genetic events that include acquisition of different mutations and/or horizontal transfer of antibiotic resistance genes. Hypermutation favours the selection of mutation-driven antibiotic resistance in P. aeruginosa strains producing chronic infections, whereas the clustering of several different antibiotic resistance genes in integrons favours the concerted acquisition of antibiotic resistance determinants. Some recent studies have shown that phenotypic resistance associated to biofilm formation or to the emergence of small-colony-variants may be important in the response of P. aeruginosa populations to antibiotics treatment.
Iron uptake in Pseudomonas
Like all aerobic bacteria, pseudomonads need to take up iron via the secretion of siderophores which complex iron (III) with high affinity. Much progress has been made in the elucidation of siderophore-mediated high-affinity iron uptake by Pseudomonas, especially in the case of the opportunistic pathogen, P. aeruginosa. Fluorescent pseudomonads produce the high-affinity peptidic siderophore pyoverdine, but also, in many cases, a second siderophore of lesser affinity for iron. Some of the genes for the biosynthesis and uptake of these siderophores have been identified and the functions of the encoded proteins known. Iron uptake via siderophores is regulated at several levels, via the general iron-sensitive repressor Fur (Ferric Uptake Regulator), via extracytoplasmic sigma factors/anti-sigma factors or via other regulators. Since pseudomonads are ubiquitous microorganisms, it is not surprising to find in their genome a large number of genes encoding receptors for the uptake of heterologous ferrisiderophores or heme reflecting their great adaptability to diverse iron sources. Another exciting development is the recent evidence for a cross-talk between the iron regulon and other regulatory networks, including the diffusible signal molecule-mediated quorum sensing in P. aeruginosa.”
PREVIOUSLY
BIODEGRADABLE PLASTICS
http://spectrevision.net/2007/06/22/biodegradable-plastics/
DOMESTICATING BIOTECH
http://spectrevision.net/2007/07/20/domesticating-biotech/
GROW YOUR OWN BACTERIAL SLAVE ARMY — SEA MONKEYS DONE FOR GOOD
http://spectrevision.net/2006/09/05/grow-your-own-bacterial-slave-army-sea-monkeys-done-for-good/