BIOMASS-FREE ETHANOL
http://nature.com/nature/journal/vaop/ncurrent/full/nature13249.html
Scientists discover how to make ethanol using just water and CO2
/ April 10, 2014
“Scientists at Stanford University in the state of California say they’ve developed a procedure for making potent liquid ethanol that doesn’t rely on corn or any other crops traditionally involved in the process. The researchers disclosed their discovery in the latest online edition of the journal Nature, and in it they say that in less than three years’ time they expect to have a prototype device ready that will make biofuel from using not much more than carbon monoxide.
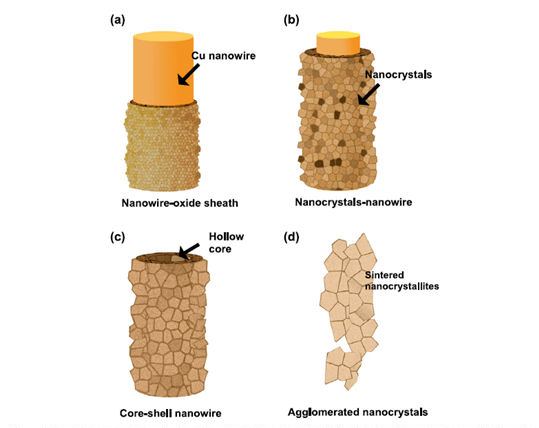
“Formation of hybrid structures: Copper oxide nanocrystals templated on ultralong copper nanowires for open network sensing at room temperature” http://www.hoghimwei.com/pdf/33.pdf
“We have discovered the first metal catalyst that can produce appreciable amounts of ethanol from carbon monoxide at room temperature and pressure – a notoriously difficult electrochemical reaction,” wrote Stanford’s Matthew Kanan, a co-author of the report released this week. The scientists say that they are still a ways from developing said prototype, but believe they are on the right track towards achieving a goal that has the potential of providing people with a new, less-costly biofuel that could essentially revamp the energy industry. “I emphasize that these are just laboratory experiments today. We haven’t built a device,” Kanan said. “But it demonstrates the feasibility of using electricity that you could get from a renewable energy source to power fuel synthesis — in this case ethanol. There are some real advantages to doing that relative to using biomass to produce ethanol.” Indeed, for one the new process unveiled this week would eliminate crops from the equation needed to make biofuel, a change that is certain to revamp America’s agriculture landscape if the resources once required were no longer needed. According to the scientists, this alone could help push food prices down by no longer diverting thousands of acres worth of key crops to the energy sector. The traditional biofuel conversion process requires a single bushel of corn, for example, in order to make just three gallons of ethanol — along with hundreds upon hundreds of gallons of water, theStanford News site reported this week.

Thin-film solar by synthesizing copper zinc tin sulfide (CZTS), selected for the lower price, environmental impact, and greater availability of the starting components
Instead, the researchers say that biofuel would be generated by using a state-of-the-art device still in development that uses two electrodes, including one made of an “oxide-derived copper,” to convert it into fuel. “Copper,” the researchers wrote in their abstract, “…is the only known material with an appreciable [carbon monoxide electroreduction activity, but in bulk form its efficiency and selectivity for liquid fuel are far too low for practical use.” But using a two-stop conversion process, the Stanford scientists say it’s entirely feasible to create a device that would create potent liquid fuel without requiring acres upon acres of farmland. “The electrochemical conversion of CO2and H2O into liquid fuel is ideal for high-density renewable energy storage and could provide an incentive for CO2capture,” they wrote. “Most materials are incapable of reducing carbon monoxide and exclusively react with water,” Kanan told the Stanford News site. “Copper is the only exception, but conventional copper is very inefficient. “Prior to our study, there was a sense that no catalyst could efficiently reduce carbon monoxide to a liquid. We have a solution to this problem that’s made of copper, which is cheap and abundant,” he added. “We hope our results inspire other people to work on our system or develop a new catalyst that converts carbon monoxide to fuel.” According to the Renewable Fuels Association, the US leads the world in ethanol production by generation around 13.3 billion gallons of the biofuel during the last calendar year.”
OXIDE-DERIVED COPPER
http://news.stanford.edu/news/2014/april/ethanol-without-plants-040914.html
Stanford scientists discover a novel way to make ethanol – without corn or other plants – from carbon monoxide gas at room temperature
by Mark Shwartz / April 9, 2014
“Stanford University scientists have found a new, highly efficient way to produce liquid ethanol from carbon monoxide gas. This promising discovery could provide an eco-friendly alternative to conventional ethanol production from corn and other crops, say the scientists. Their results are published in the April 9 advanced online edition of the journal Nature. “We have discovered the first metal catalyst that can produce appreciable amounts of ethanol from carbon monoxide at room temperature and pressure – a notoriously difficult electrochemical reaction,” said Matthew Kanan, an assistant professor of chemistry at Stanford and coauthor of the Naturestudy. Most ethanol today is produced at high-temperature fermentation facilities that chemically convert corn, sugarcane and other plants into liquid fuel. But growing crops for biofuel requires thousands of acres of land and vast quantities of fertilizer and water. In some parts of the United States, it takes more than 800 gallons of water to grow a bushel of corn, which, in turn, yields about 3 gallons of ethanol. The new technique developed by Kanan and Stanford graduate student Christina Li requires no fermentation and, if scaled up, could help address many of the land- and water-use issues surrounding ethanol production today. “Our study demonstrates the feasibility of making ethanol by electrocatalysis,” Kanan said. “But we have a lot more work to do to make a device that is practical.”
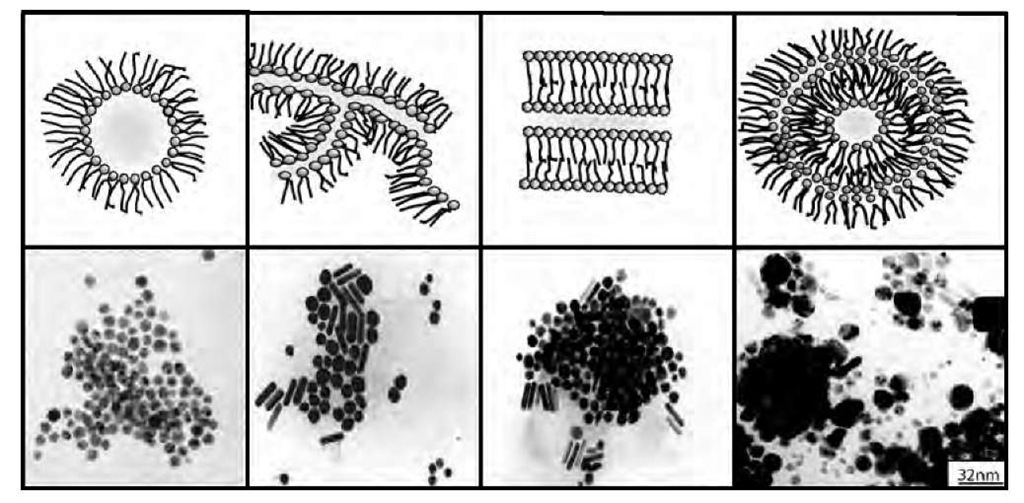
Change in the shape of copper nanocrystals in various colloidal solutions differing by their structures. In lamellar phase obtained at w =11, a mixture of spheres and cylinders is produced, whereas at w = 30, supra-aggregates are formed and a large variety of copper nanocrystals differing by their shapes are produced.

TEM images at medium and high resolution showing different shapes and structures of copper nanocrystals. At left: A, B-decahedron; C, D, E, F truncated decahedrons. At right f.c.c. copper cubes
Novel electrodes
Two years ago, Kanan and Li created a novel electrode made of a material they called oxide-derived copper. They used the term “oxide-derived” because the metallic electrode was produced from copper oxide. “Conventional copper electrodes consist of individual nanoparticles that just sit on top of each other,” Kanan said. “Oxide-derived copper, on the other hand, is made of copper nanocrystals that are all linked together in a continuous network with well-defined grain boundaries. The process of transforming copper oxide into metallic copper creates the network of nanocrystals.” For the Nature study, Kanan and Li built an electrochemical cell – a device consisting of two electrodes placed in water saturated with carbon monoxide gas. When a voltage is applied across the electrodes of a conventional cell, a current flows and water is converted to oxygen gas at one electrode (the anode) and hydrogen gas at the other electrode (the cathode). The challenge was to find a cathode that would reduce carbon monoxide to ethanol instead of reducing water to hydrogen. “Most materials are incapable of reducing carbon monoxide and exclusively react with water,” Kanan said. “Copper is the only exception, but conventional copper is very inefficient.” In the Nature experiment, Kanan and Li used a cathode made of oxide-derived copper. When a small voltage was applied, the results were dramatic. “The oxide-derived copper produced ethanol and acetate with 57 percent faradaic efficiency,” Kanan said. “That means 57 percent of the electric current went into producing these two compounds from carbon monoxide. We’re excited because this represents a more than 10-fold increase in efficiency over conventional copper catalysts. Our models suggest that the nanocrystalline network in the oxide-derived copper was critical for achieving these results.”
Carbon neutral
The Stanford team has begun looking for ways to create other fuels and improve the overall efficiency of the process. “In this experiment, ethanol was the major product,” Kanan said. “Propanol would actually be a higher energy-density fuel than ethanol, but right now there is no efficient way to produce it.” In the experiment, Kanan and Li found that a slightly altered oxide-derived copper catalyst produced propanol with 10 percent efficiency. The team is working to improve the yield for propanol by further tuning the catalyst’s structure. Ultimately, Kanan would like to see a scaled-up version of the catalytic cell powered by electricity from the sun, wind or other renewable resource. For the process to be carbon neutral, scientists will have to find a new way to make carbon monoxide from renewable energy instead of fossil fuel, the primary source today. Kanan envisions taking carbon dioxide (CO2) from the atmosphere to produce carbon monoxide, which, in turn, would be fed to a copper catalyst to make liquid fuel. The CO2 that is released into the atmosphere during fuel combustion would be re-used to make more carbon monoxide and more fuel – a closed-loop, emissions-free process. “Technology already exists for converting CO2 to carbon monoxide, but the missing piece was the efficient conversion of carbon monoxide to a useful fuel that’s liquid, easy to store and nontoxic,” Kanan said. “Prior to our study, there was a sense that no catalyst could efficiently reduce carbon monoxide to a liquid. We have a solution to this problem that’s made of copper, which is cheap and abundant. We hope our results inspire other people to work on our system or develop a new catalyst that converts carbon monoxide to fuel.”
PREVIOUSLY
“MIT chemists Daniel Nocera and Matthew Kanan have discovered a way to to convert sunlight into chemical fuel by using Cobalt to split water into oxygen and hydrogen”
HARNESSING LIGHTNING
http://spectrevision.net/2010/08/26/harnessing-lightning/



