
Seen through an electron microscope, the Desulfobulbaceae — the researchers haven’t yet given them a genus or species name — appear in blue. They link end-to-end, forming filaments nearly an inch in length. {Image: Nils Risgaard-Petersen}
EXPLAINS CRACKLE
http://phys.org/news/2014-08-bacterial-nanowires-thought.html
http://scitech.au.dk/en/current-affairs/news/show/artikel/living-cables-explain-enigmatic-electric-currents/
The enigma of electric currents in the seabed is solved. Scientists from Aarhus University have discovered bacteria that function as living electrical cables. Each of the centimetre-long ‘cable bacteria’ contains a bundle of insulated wires leading an electric current from one end to the other. Electricity and seawater are usually a bad mix. And it was thus a very big surprise when scientists from Aarhus University a few years ago discovered electric currents between biological processes in the seabed. Since then they have been searching for an explanation and together with partners from the University of Southern California, USA, they now present sensational results in Nature.
“Our experiments showed that the electric connections in the seabed must be solid structures built by bacteria,” says PhD student Christian Pfeffer, Aarhus University. He could interrupt the electric currents by pulling a thin wire horizontally through the seafloor. Just as when an excavator cuts our electric cables. In microscopes, scientists found a hitherto unknown type of long, multi-cellular bacteria that was always present when scientists measured the electric currents. “The incredible idea that these bacteria should be electric cables really fell into place when, inside the bacteria, we saw wire-like strings enclosed by a membrane,” says Nils Risgaard-Petersen, Aarhus University.
SQUARE KILOMETERS of LIVING CABLES
The bacterium is one hundred times thinner than a hair and the whole bacterium functions as an electric cable with a number of insulated wires within it. Quite similar to the electric cables we know from our daily lives. “Such unique insulated biological wires seem simple but with incredible complexity at nanoscale,” says PhD student Jie Song, Aarhus University, who used nanotools to map the electrical properties of the cable bacteria. In an undisturbed seabed more than tens of thousands kilometers cable bacteria live under a single square meter seabed. The ability to conduct an electric current gives cable bacteria such large benefits that it conquers much of the energy from decomposition processes in the seabed.
Unlike all other known forms of life, cable bacteria maintain an efficient combustion down in the oxygen-free part of the seabed. It only requires that one end of the individual reaches the oxygen which the seawater provides to the top millimeters of the seabed. The combustion is a transfer of the electrons of the food to oxygen which the bacterial inner wires manage over centimeter-long distances. However, small disturbances can lead to fatal “cable breakage” in the fragile bacteria. “On the one hand, it is still very unreal and fantastic. On the other hand, it is also very tangible,” says Professor at Aarhus University, Lars Peter Nielsen, who is in charge of exploring the natural electrical currents. Along with a number of international cooperation partners, several scientists at Aarhus University already address the new and exciting questions that arise. Right from the understanding of bioelectronics at the molecular level to the role of cable bacteria in the history of Earth. The future will tell whether this wondrous result of the biological evolution can also be used in new types of electronics.

In the photo above, orange strands of the new Desulfobulbaceae stretch in a laboratory beaker between a reddish, oxygen-rich sediment layer and a dark, sulfurous, oxygen-depleted layer. This is the essential structure of much of the world’s seafloor, and one that the new Desulfobulbaceae evolved to exploit: One end feeds on hydrogen sulfide below, pulling out an electron that’s sent up the chain and, at the other end, is used to pull in oxygen. It’s a simple form of breathing. Water is generated as a byproduct. {Images: Nils Risgaard-Petersen}
NEW (OXYGEN-FREE) GENUS
http://www.wired.com/wiredscience/2012/10/bacteria-electric-wires/
Electric Bugs: New Microbe Forms Living, Deep-Sea Power Cables
by Brandon Keim / October 24, 2012
The world’s deep sea floors are dark and airless places, but vast swaths may pulse gently with energy conducted through a type of newly discovered bacteria that forms living electrical cables.The bacteria were first detected in 2010 by researchers perplexed at chemical fluctuations in sediments from the bottom of Aarhus Bay in Denmark. Almost instantaneously linking changing oxygen levels in water with reactions in mud nearly an inch below, the fluctuations occurred too fast to be explained by chemistry. Only an electrical signal made sense — but no known bacteria could transmit electricity across such comparatively vast distances. Were bacteria the size of humans, the signals would be making a journey 12 miles long. Now the mysterious bacteria have been identified. They belong to a microbial family called Desulfobulbaceae, though they share just 92 percent of their genes with any previously known member of that family. They deserve to be considered a new genus, the study of which could open a new scientific frontier for understanding the interface of biology, geology and chemistry across the undersea world. The bacteria are described Oct. 24 in Nature by researchers led by microbiologists Christian Pfeffer, Nils Risgaard-Petersen and Lars Peter Nielsen of Aarhus University. On the following pages, Wired takes a look at these marvelous microbes.

{Image: Karen E. Thomsen}
The new Desulfobulbaceae, seen in cross-section above, has a shape seemingly adapted to conducting electricity. Down each bacterium run deep channels, which are aligned continuously as the bacteria join into one long filament. It’s through these channels that electrons likely course. (This is still speculation, albeit informed — the bacteria clearly transfer electrons, but the exact route hasn’t been mapped.) The walls of the channels and a surrounding membrane may have insulating properties, like sheathing around a wire.

{Image: Mingdong Dong}
A Single Organism: Each filament of the new Desulfobulbaceae doesn’t merely represent the end-to-end alignment of many individual microbes, but should be a considered a single multicellular organism, said Nielsen.

{Image: Nils Risgaard-Petersen}
In just one teaspoon of mud, the researchers found a full half-mile of Desulfobulbaceae cable, and it’s not just a Danish phenomenon. Nielsen said other researchers have sent him samples from seafloors around the world, including Tokyo Bay. It’s possible that, at the microbial level, the deep seafloor is humming with current. With so much electricity being transferred, are other organisms tapping the lines? Might the Desulfobulbaceae be a power source for entire as-yet-unappreciated deep-sea microbial ecologies, which in turn shape some of the planet’s fundamental biogeochemical processes? That’s “an interesting possibility,” said Nielsen, but it’s still speculation. Less speculatively, the Desulfobulbaceae are definitely breaking down iron sulfides and carbonates in deeper sediment, while generating iron oxide and magnesium calcite at the surface, Nielsen said. The latter are important compounds for life in the oceans above, and ultimately on land. If the new Desulfobulbaceae are as widespread and populous as they seem, they could be an important component of life’s deep-time cycles.

Study co-author Christian Pfeffer pulls a thin wire through the seabed, severing the electrical connections. {Image: Nils Risgaard-Petersen}
What Next?
In addition to ecological questions, the researchers next want to learn more about the new Desulfobulbaceae’s structure and physiology. “These are exciting times for microbiologists,” wrote microbiologist Gemma Reguera of Michigan State University in a commentary accompanying the new study. “The present work reminds us — one more time — just how much more awaits discovery.”
{Citations: “Filamentous bacteria transport electrons over centimetre distances.” Christian Pfeffer, Steffen Larsen, Jie Song, Mingdong Dong, Flemming Besenbacher, Rikke Louise Meyer, Kasper Urup Kjeldsen, Lars Schreiber, Yuri A. Gorby, Mohamed Y. El-Naggar, Kar Man Leung, Andreas Schramm, Nils Risgaard-Petersen & Lars Peter Nielsen. Nature, Vol. 490, No. 7421, 25 Oct. 2012.
“Bacterial power cords.” By Gemma Reguera. Nature, Vol. 490, No. 7421, 25 Oct. 2012}

Image: At left, Nielsen measures current in the sediment sample; at right, a close-up view of the sediment. {Image: Nils Risgaard-Petersen}
BIOELECTRICAL ECOLOGY
http://www.wired.com/wiredscience/2010/02/electric-ocean-bacteria/
Deep-Sea Bacteria Form Avatar-Style Electrochemical Networks
by Brandon Keim / February 25, 2010
According to findings, bacteria appear to conduct electrical currents across the ocean floor, driving linked chemical reactions at relatively vast distances. Noticed only when reseachers happened to test sediment leftovers from another experiment, the phenomenon may add a new mechanism to Earth’s biogeochemistry. “The cycling of elements and life at the bottom of the sea, and in soil, and anywhere else you’re short of oxygen — this could help us understand those processes,” said microbiologist Lars Peter Nielsen of Denmark’s Aarhus University, co-author of the study, published Feb. 24 in Nature.
The original focus of Nielsen’s team wasn’t seafloor conductivity, but an especially interesting species of sulfur bacteria found on the floor of Aarhus Bay. To help quantify their chemical activity, the researchers kept a few beakers of seawater and sulfur bacteria-free sediment for comparison. After those experiments ended, the beakers were almost forgotten. Then, a few weeks later, the researchers noticed strange patterns of activity. Changing oxygen levels in water above the top sediment layer were almost immediately followed by chemical fluctuations several layers down. The distance was so great, and the response time so quick, that usual methods of chemical transport — molecular diffusion, or a slow drift from high to low concentration — couldn’t explain it. At first, the researchers were stumped. Then they realized the process made sense if bacteria in the top and bottom layers were linked. Anything that affected oxygen-processing bacteria up top would also affect the sulfide-eating microbes below. It would explain the apparent connection; and an electrical linkage would explain the speed. It would also boggle the mind.
“Such hypotheses would at one time have been considered heretical,” wrote Kenneth Nealson, a University of Southern California microbiologist, in an accompanying commentary in Nature. A half-inch gap “doesn’t seem like much of a distance. But to a bacterium it amounts to 10,000 body lengths, equivalent to about 20 kilometers (12 miles) in human terms.” In recent years, however, scientists have found species of microbes with outer membranes covered by electron-transporting enzymes, or studded with conductive, micrometer-scale filaments. These are used in driving experimental microbial fuel cells, and are known to be found in the Aarhus Bay mud. Those sediments also contain trace amounts of pyrite, an electrically conductive mineral. The top sediment layer also had a low concentration of hydrogen ions, something that could only be explained through an electrochemical reaction, with electrons conducted from a distance, said Nielsen.
Nealson called the findings “astonishing,” and said they “may be relevant to energy transfer and electron flow through many different environments.” They could eventually applied to bacteria-based schemes for bioremediation, carbon sequestration and energy production.
Citations: “Electric currents couple spatially separated biogeochemical processes in marine sediment.” By Lars Peter Nielsen, Nils Risgaard-Petersen, Henrik Fossing, Peter Bondo Christensen & Mikio Sayam. Nature, Vol. 463, No. 7284, February 25, 2010.
“Sediment reactions defy dogma.” By Kenneth H. Nealson. Nature, Vol. 463, No. 7284, February 25, 2010.
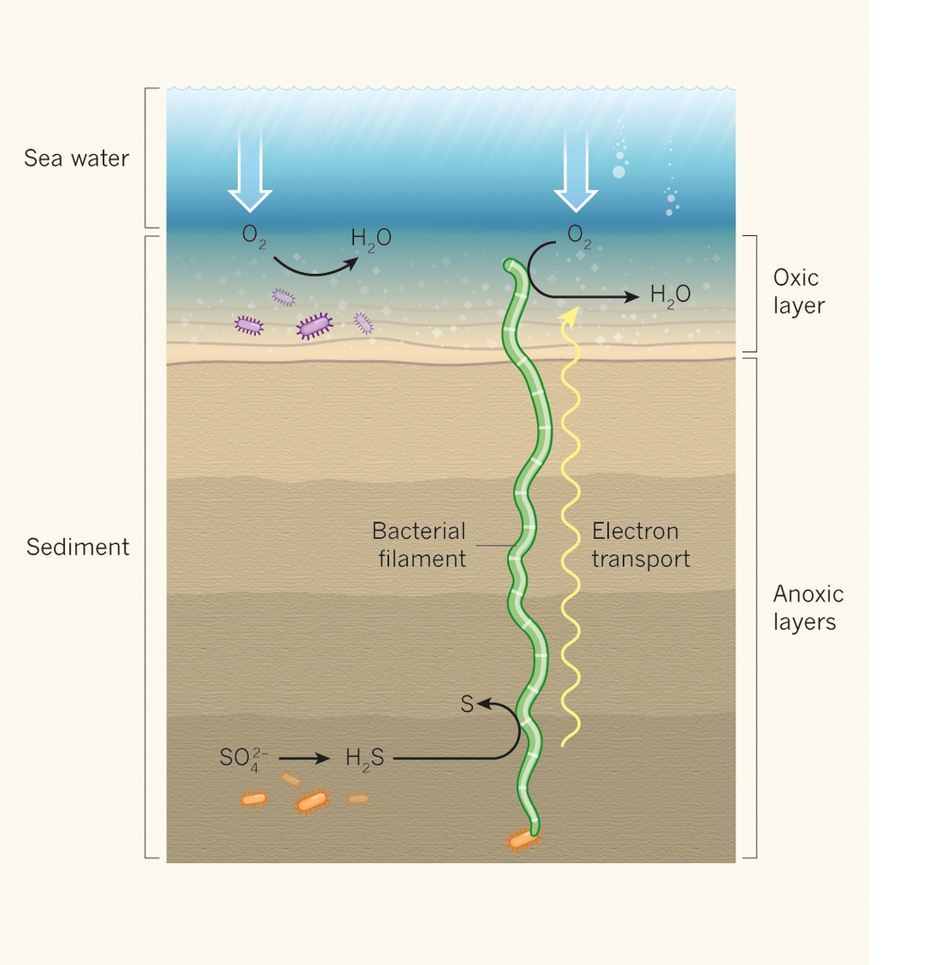
Microorganisms (purple) in the upper layers of marine sediments use oxygen (O2) that diffuses from sea water as an acceptor of electrons, which they produce in energy-generating metabolic reactions. As a result, other microbes (orange) in deeper, anoxic layers (where oxygen is scarce or absent) have to use other electron acceptors such as sulphate (SO42−) for growth. Transfer of electrons to oxygen results in the formation of water, whereas electron transfer to sulphate produces hydrogen sulphide (H2S), which is poisonous to many organisms. Pfeffer et al. provide evidence that long bacterial filaments could transport electrons generated when hydrogen sulphide is converted into sulphur (S) at the bottom of the sediments and use them to consume oxygen in the upper layers. {Image: Gemma Reguera/Nature}
LIVING POWER CABLES
http://www.kurzweilai.net/living-power-cables-discovered
Multicellular bacteria transmit electrons across relatively enormous distances / October 26, 2012
A multinational research team has discovered filamentous bacteria that function as living power cables that transmit electrons thousands of cell lengths away. The Desulfobulbus bacterial cells, which are only a few hundreds of a nanometer long each, are so tiny that they are invisible to the naked eye. And yet, under the right circumstances, they form a multicellular filament that can transmit electrons across a distance as large as 1 centimeter as part of the filament’s respiration and ingestion processes. “To move electrons over these enormous distances in an entirely biological system would have been thought impossible,” said Moh El-Naggar, assistant professor of physics at the USC Dornsife College of Letters, Arts and Sciences, and co-author of the Nature paper.
Aarhus University scientists had discovered a seemingly inexplicable electric current on the sea floor years ago. The new experiments revealed that these currents are mediated by a hitherto unknown type of long, multicellular bacteria that act as living power cables “Until we found the cables we imagined something cooperative where electrons were transported through external networks between different bacteria. It was indeed a surprise to realize, that it was all going on inside a single organism,” said Lars Peter Nielsen of the Aarhus Department of Bioscience, and a corresponding author of the Nature paper.

SEM image of four filamentous_Desulfobulbaceae bacteria cells {Image: Christian Pfeffer et al./Nature}
The team studied bacteria living in marine sediments that power themselves by oxidizing hydrogen sulfide. Cells at the bottom live in a zone that is poor in oxygen but rich in hydrogen sulfide, and those at the top live in an area rich in oxygen but poor in hydrogen sulfide. The solution? They form long chains that transport individual electrons from the bottom to the top, completing the chemical reaction and generating life-sustaining energy. “You have feeder cells on one end and breather cells on the other, allowing the whole living cable to survive,” El-Naggar said. Aarhus and USC researchers collaborated to use physical techniques to evaluate the long-distance electron transfer in the filamentous bacteria. El-Naggar and his colleagues had previously used scanning-probe microscopy and nanofabrication methods to describe how bacteria use nanoscale structures called “bacterial nanowires” to transmit electrons many body lengths away from cells. This research was funded by European Research Council, the Danish National Research Foundation, the Danish Foundation for Independent Research and the German Max Planck Society.
References
Gemma Reguera, Microbiology: Bacterial power cords, Nature, 2012, DOI: 10.1038/nature11638
Christian Pfeffer et al., Filamentous bacteria transport electrons over centimetre distances, Nature, 2012, DOI: 10.1038/nature11586

http://www.bme.duke.edu/news/3079
BIOELECTRIC BIOMIMICRY
http://www.telegraph.co.uk/science/roger-highfield/9627744/The-electrical-storm-gathering-in-biology.html
The electrical storm gathering in biology
by Roger Highfield / 23 Oct 2012
Today, biologists are realising that electricity is even more important than was hitherto thought – so much so that some are talking about a new bioelectrical revolution. It not only governs the contraction of our muscles and carries impulses through our nerves, but also holds the key to a host of illnesses, from the most intense migraines to cystic fibrosis. Last week, at a lecture in the Royal Institution, London, the Oxford University physiologist Frances Ashcroft explained how this revolution in bioelectricity has happened. While the electricity we use to power motors, make lights shine and bring our computers to life relies on electrons – the fundamental sub-atomic particles which carry electrical charge – the electricity in our bodies is carried by larger, more complex charged atoms, or ions, which are found in salts such as sodium chloride. While electricity in wires travels at the speed of light, (around 186,000 miles per second), electrical signals are carried around our bodies at a far slower (if still rapid) half a mile per second, or about 1800 mph.
As bioelectricity flashes in and out of our cells, it generates currents of a few picoamperes – about a hundred billionth of the current that makes a light bulb glow. Somehow, the ions carrying these currents have to find a way past the insulating greasy membrane that protects the watery contents of every cell. The realisation that cell membranes are studded with tiny pores (constructed from specific proteins), which allow the free movement of ions, dates back to in the 1950s and the pioneering studies of Alan Hodgkin and Andrew Huxley in Cambridge and Bernard Katz at University College London. These pores are ion channels, and they regulate all life, from the moment of conception until we draw our last breath. Indeed, according to Ashcroft, these channels are truly the “spark of life” – the title of her recent book on the subject. From the lashing of the sperm’s tail to the beating of our hearts, the craving for yet another chocolate, or the feel of the sun on your skin, everything is underpinned by ion channel activity.
In 1984, Ashcroft discovered an ion channel through which potassium ions leave cells and observed that it was closed by the breakdown of glucose, triggering the release of insulin. She was so excited that she did not sleep; the next morning, thought she had made a mistake. She hadn’t. Two decades later it was found that a rare childhood form of diabetes resulted from a defect in this channel and, in a remarkable twist, could be treated by taking pills called sulphonylureas, initially trialled as a treatment for typhoid. It turns out that faulty ion channels are actually responsible for a remarkably wide range of human and animal diseases. Pigs that shiver themselves to death, myotonic goats that stiffen so much they topple over when startled, humans with cystic fibrosis, epilepsy, heart arrhythmias or migraine – all are victims of ion channel dysfunction. Mutations in sodium channels, for example, underlie inherited forms of epilepsy (when an electrical storm erupts in the brain), migraine headaches, heart rhythm disturbances, paralysis, and some chronic pain syndromes. In the past few years, important clues to understanding what goes wrong, and how this can be fixed, have come from working out what ion channels look like.
Sodium channels – which allow sodium ions to pass – are found in “excitable” cells such as the neurons in your brain, or the cells found in heart muscle, or the nerve cells that carry signals of pain, hot or cold. Their atomic structure was only solved last year, by William Catterall’s team from the University of Washington in Seattle. Many drugs work by interacting with ion channels and knowing the shape of the protein, and what the drug-binding sites look like, is expected to stimulate the design of new chemicals, which can alter the protein’s structure. Poisons target ion channels, too. In the past few days, Sylvie Diochot and Anne Baron from the French National Centre for Scientific Research (CNRS) have reported a remarkable discovery. The venom of the black mamba, one of the world’s nastiest snakes, contains chemicals that block nerve ion channels. These chemicals, mambalgins, stop a specific type of ion channel found in pain cells from opening. By doing so, they relieve pain as effectively as morphine, without its side effects. Thanks to the second electrical revolution, expect a new generation of drugs to fine-tune the electrical workings of your heart, nerves and brain.
