
“orb webs illustrating web architecture and debris decoration”
eDNA FUNGAL SAMPLING from SPIDERWEBS
https://sciencedirect.com/S2589004224001251
https://phys.org/spiders-biologists-artificial-webs
https://phys.org/spider-silk-optimized-cell-specific
https://phys.org/spider-webs-capture-hidden-fungal
Spider webs capture hidden fungal diversity in Thai rice fields
by┬ĀPensoft Publishers┬Ā /┬Ā June 3, 2026
“A new study┬Āpublished in the open-access┬ĀBiodiversity Data Journal suggests that spider websŌĆöparticularly those incorporating environmental debrisŌĆöcan serve as natural, non-destructive collectors of fungal material in agricultural ecosystems. The findings show that viable fungi can be recovered from these structures, including lineages that may represent previously undocumented diversity. “Spider webs are often overlooked structures in the environment, yet they can function as natural collectors of biological material,” said Thanakron Into, a student at Thammasat University. “Our findings suggest that they can be used as a complementary approach to access microbial communities without disturbing the surrounding ecosystem.”
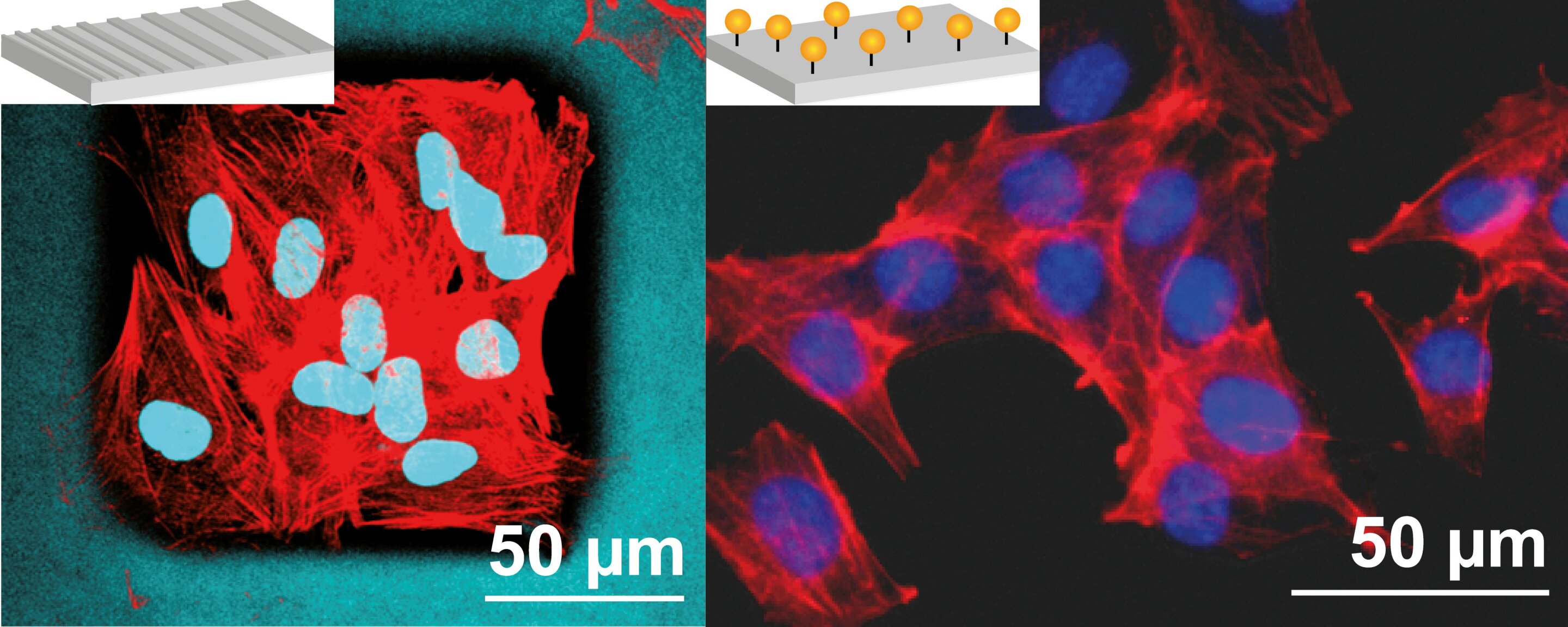
“Left: Skin fibroblasts on structured coatings of spider silk; right: muscle progenitor cells on a scaffold of peptide-modified spider silk. Red: actin cytoskeleton, turquoise: nucleus & spider silk scaffold, blue: nucleus.”
Researchers from Thammasat University and the National Center for Genetic Engineering and Biotechnology (BIOTEC), Thailand, investigated whether the adhesive and┬Āparticle-trapping properties of spider silk could be used to capture and culture fungi associated with airborne and environmental particles. Unlike DNA-based methods, which detect genetic material regardless of viability, this approach allows for the recovery of living organisms that can be further studied. The study focused on tropical rice fields, using webs of the orb-weaving spider Cyclosa mulmeinensis, a species known for constructing distinctive “trashline” decorationsŌĆölinear accumulations of plant fragments, insect remains, and other debris within the web. These structures can intercept a variety of particles, some of which may carry fungal propagules.

“Representative orb webs of Cyclosa mulmeinensis
illustrating web architecture and debris decoration”
Webs were collected from rice-field embankments in Pathum Thani, Nakhon Nayok, and Phetchaburi provinces using sterile techniques. In the laboratory, material retained on the silk was gently removed and cultured, yielding 112 viable fungal isolates. These isolates were grouped into 23 taxa across six genera, including Alternaria, Aspergillus, Cladosporium, Fusarium, Penicillium, and Talaromyces. “We were particularly surprised that many of the fungi recovered from the webs remained viable and could be cultured,” Into noted. “This enables further investigation beyond presence or absence, including their biological characteristics.” Some genetic lineages ŌĆö particularly within Cladosporium and Talaromyces ŌĆö did not match currently described species in available databases, indicating that additional, undocumented diversity may be present in these systems.
Conventional approaches to fungal monitoring typically rely on soil, air, or plant sampling, or on molecular methods that may not distinguish between living and non-living material. In this context, spider webs may provide a useful┬Āsupplementary sampling surface for capturing biologically relevant particles. Because spider webs are naturally maintained and, in some species, periodically rebuilt, this method can be applied with minimal disturbance to both the organisms and their environment. Importantly, the spiders themselves were not harmed during sampling, as only small sections of the web were collected.

“Artificial spiderwebs are as good as natural
spiderwebs at capturing DNA from the air”
“The ability to recover living fungi from these naturally occurring structures adds a practical dimension to biodiversity studies,” Into added. “It provides a way to link environmental sampling with downstream biological work.” The idea that something as familiar as a spider web could quietly capture a hidden layer of biodiversity highlights how much of the natural world remains overlooked in plain sight. While further work is needed to evaluate how broadly this approach can be applied, the study highlights the potential of spider webs as an additional tool for exploring microbial diversity in agricultural landscapes.”
Publication details
Thanakron Into et al, Spider webs as reservoirs of culturable fungal diversity: evidence from orb-weaving Cyclosa mulmeinensis spider in Thai rice agroecosystems,┬ĀBiodiversity Data Journal┬Ā(2026).┬ĀDOI: 10.3897/bdj.14.e187035
PREVIOUSLY
FLYING ELECTRIC SPIDERS
https://spectrevision.net/2018/09/04/flying-electric-spiders/
PALEOGENETICS
https://spectrevision.net/2022/12/23/paleogenetics/
ENVIRONMENTAL DNA
https://spectrevision.net/2026/04/30/environmental-dna/
