LIVING to 1000
http://en.wikipedia.org/wiki/Aubrey_de_Grey
http://news.bbc.co.uk/2/hi/uk_news/4003063.stm
‘We will be able to live to 1,000’
by Dr Aubrey de Grey / 3 December, 2004
“Life expectancy is increasing in the developed world. But Cambridge University geneticist Aubrey de Grey believes it will soon extend dramatically to 1,000. Here, he explains why. Ageing is a physical phenomenon happening to our bodies, so at some point in the future, as medicine becomes more and more powerful, we will inevitably be able to address ageing just as effectively as we address many diseases today. I claim that we are close to that point because of the SENS (Strategies for Engineered Negligible Senescence) project to prevent and cure ageing. It is not just an idea: it’s a very detailed plan to repair all the types of molecular and cellular damage that happen to us over time. And each method to do this is either already working in a preliminary form (in clinical trials) or is based on technologies that already exist and just need to be combined.
This means that all parts of the project should be fully working in mice within just 10 years and we might take only another 10 years to get them all working in humans. When we get these therapies, we will no longer all get frail and decrepit and dependent as we get older, and eventually succumb to the innumerable ghastly progressive diseases of old age. We will still die, of course – from crossing the road carelessly, being bitten by snakes, catching a new flu variant etcetera – but not in the drawn-out way in which most of us die at present. So, will this happen in time for some people alive today? Probably. Since these therapies repair accumulated damage, they are applicable to people in middle age or older who have a fair amount of that damage. I think the first person to live to 1,000 might be 60 already. It is very complicated, because ageing is. There are seven major types of molecular and cellular damage that eventually become bad for us – including cells being lost without replacement and mutations in our chromosomes. Each of these things is potentially fixable by technology that either already exists or is in active development.
‘Youthful not frail’
The length of life will be much more variable than now, when most people die at a narrow range of ages (65 to 90 or so), because people won’t be getting frailer as time passes. The average age will be in the region of a few thousand years. These numbers are guesses, of course, but they’re guided by the rate at which the young die these days. If you are a reasonably risk-aware teenager today in an affluent, non-violent neighbourhood, you have a risk of dying in the next year of well under one in 1,000, which means that if you stayed that way forever you would have a 50/50 chance of living to over 1,000. And remember, none of that time would be lived in frailty and debility and dependence – you would be youthful, both physically and mentally, right up to the day you mis-time the speed of that oncoming lorry.
Should we cure ageing?
Curing ageing will change society in innumerable ways. Some people are so scared of this that they think we should accept ageing as it is. I think that is diabolical – it says we should deny people the right to life. The right to choose to live or to die is the most fundamental right there is; conversely, the duty to give others that opportunity to the best of our ability is the most fundamental duty there is. There is no difference between saving lives and extending lives, because in both cases we are giving people the chance of more life. To say that we shouldn’t cure ageing is ageism, saying that old people are unworthy of medical care.
Playing God?
People also say we will get terribly bored but I say we will have the resources to improve everyone’s ability to get the most out of life. People with a good education and the time to use it never get bored today and can’t imagine ever running out of new things they’d like to do. And finally some people are worried that it would mean playing God and going against nature. But it’s unnatural for us to accept the world as we find it. Ever since we invented fire and the wheel, we’ve been demonstrating both our ability and our inherent desire to fix things that we don’t like about ourselves and our environment. We would be going against that most fundamental aspect of what it is to be human if we decided that something so horrible as everyone getting frail and decrepit and dependent was something we should live with forever. If changing our world is playing God, it is just one more way in which God made us in His image.”
CONTACT
Aubrey de Grey
http://sens.org/index.php?pagename=mj_about_who_founders#adg
http://ted.com/speakers/aubrey_de_grey.html
email : aubrey [at] sens [dot] org
LONGEVITY ESCAPE VELOCITY
http://edge.org/3rd_culture/degrey07/degrey07_index.html
Bootstrapping Our Way To An Ageless Future
by Aubrey de Grey / 2007
“An important fact is that the therapies we develop in a decade or so in mice, and those that may come only a decade or two later for humans, will not be perfect. Other things being equal, there will be a residual accumulation of damage within our bodies, however frequently and thoroughly we apply these therapies, and we will eventually experience age-related decline and death just as now, only at a greater age. Probably not all that much greater either—probably only 30-50 years older than today. But other things won’t be equal, and I’m going to explain why not—and why, as you may already know from other sources, I expect many people alive today to live to 1000 years of age and to avoid age-related health problems even at that age. I’ll start by describing why it’s unrealistic to expect these therapies to be perfect.
Evolution didn’t leave notes
The body is a machine, and that’s both why it ages and why it can in principle be maintained. I have made a comparison with vintage cars, which are kept fully functional even 100 years after they were built, using the same maintenance technologies that kept them going 50 years ago when they were already far older than they were ever designed to be. More complex machines can also be kept going indefinitely, though the expense and expertise involved may mean that this never happens in practice because replacing the machine is a reasonable alternative. This sounds very much like a reason to suppose that the therapies we develop to stave off aging for a few decades will indeed be enough to stave it off indefinitely.
But actually that’s overoptimistic. All we can reliably infer from a comparison with man-made machines is that a truly comprehensive panel of therapies, which truly repairs everything that goes wrong with us as a result of aging, is possible in principle—not that it is foreseeable. And in fact we can see that actually one thing about them is very unlike maintenance of a man-made machine: these therapies strive to minimally alter metabolism itself, and target only the initially inert side-effects of metabolism, whereas machine maintenance may involve adding extra things to the machinery itself (to the fuel or the oil of a car, for example). We can get away with this sort of invasive maintenance of man-made machines because we (well, some of us!) know how they work right down to the last detail, so we can be adequately sure that our intervention won’t have unforeseen side-effects. With the body—even the body of a mouse—we are still profoundly ignorant of the details, so we have to sidestep our ignorance by interfering as little as possible.
What that means for efficacy of therapies is that, as we fix more and more aspects of aging, you can bet that new aspects will be unmasked. These new things will not be fatal at a currently normal age, because if they were, we’d know about them already. But they’ll be fatal eventually, unless we work out how to fix them too. Even within each existing category, there are some subcategories that will be easier to fix than others. For example, there are lots of chemically distinct cross-links responsible for stiffening our arteries; some of them may be broken with ALT-711 and related molecules, but others will surely need more sophisticated agents that have not yet been developed. Another example: obviating mitochondrial DNA by putting modified copies of it into the cell’s chromosomes requires gene therapy, and thus far we have no gene therapy delivery system (“vector”) that can safely get into all cells, so for the foreseeable future we’ll probably only be able to protect a subset of cells from mtDNA mutations. Much better vectors will be needed if we are to reach all cells.
In practice, therefore, therapies that rejuvenate 60-year-olds by 20 years will not work so well the second time around. When the therapies are applied for the first time, the people receiving them will have 60 years of “easy” damage (the types that the therapies can remove) and also 60 years of “difficult” damage. But by the time beneficiaries of these therapies have returned to biologically 60 (which, let’s presume, will happen when they’re chronologically about 80), the damage their bodies contain will consist of 20 years of “easy” damage and 80 years of “difficult” damage. Thus, the therapies will only rejuvenate them by a much smaller amount, say ten years. So they’ll have to come back sooner for the third treatment, but that will benefit them even less… and very soon, just like Achilles catching up with the tortoise in Zeno’s paradox, aging will get the better of them. An extremely counterintuitive fact is that, even though it will be much harder to double a middle-aged human’s remaining lifespan than a middle-aged mouse’s, multiplying that remaining lifespan by much larger factors—ten or 30, say—will be much easier in humans than in mice.
The two-speed pace of technology
I’m now going to switch briefly from science to the history of science, or more precisely the history of technology. It was well before recorded history that people began to take an interest in the possibility of flying: indeed, this may be a desire almost as ancient as the desire to live forever. Yet, with the notable but sadly unreproduced exception of Daedalus and Icarus, no success in this area was achieved until about a century ago. (If we count balloons then we must double that, but really only airships—balloons that can control their direction of travel reasonably well—should be counted, and they only emerged at around the same time as the aircraft.) Throughout the previous few centuries, engineers from Leonardo on devised ways to achieve controlled powered flight, and we must presume that they believed their designs to be only a few decades (at most) from realisation. But they were wrong.
Ever since the Wright brothers flew at Kitty Hawk, however, things have been curiously different. Having mastered the basics, aviation engineers seem to have progressed to ever greater heights (literally as well as metaphorically!) at an almost serenely smooth pace. To pick a representative selection of milestones: Lindbergh flew the Atlantic 24 years after the first powered flight occurred, the first commercial jetliner (the Comet) debuted 22 years after that, and the first supersonic airliner (Concorde) followed after a further 20 years.
This stark contrast between fundamental breakthroughs and incremental refinements of those breakthroughs is, I would contend, typical of the history of technological fields. Further, I would argue that it’s not surprising: both psychologically and scientifically, bigger advances are harder to estimate the difficulty of.
I mention all this, of course, because of what it tells us about the likely future progress of life extension therapies. Just as people were wrong for centuries about how hard it is to fly but eventually cracked it, we’ve been wrong since time immemorial about how hard aging is to combat but we’ll eventually crack it too. But just as people have been pretty reliably correct about how to make better and better aircraft once they had the first one, we can expect to be pretty reliably correct about how to repair the damage of aging more and more comprehensively once we can do it a little. That’s not to say it’ll be easy, though. It’ll take time, just as it took time to get from the Wright Flyer to Concorde. And that is why, if you want to live to 1000, you can count yourself lucky that you’re a human and not a mouse. Let me take you through the scenario, step by step.
Suppose we develop Robust Mouse Rejuvenation in 2016, and we take a few dozen two-year-old mice and duly treble their one-year remaining lifespans. That will mean that, rather than dying in 2017 as they otherwise would, they’ll die in 2019. Well, maybe not—in particular, not if we can develop better therapies by 2018 that re-treble their remaining lifespan (which will by now be down to one year again). But remember, they’ll be harder to repair the second time: their overall damage level may be the same as before they received the first therapies, but a higher proportion of that damage will be of types that those first therapies can’t fix. So we’ll only be able to achieve that re-trebling if the therapies we have available by 2018 are considerably more powerful than those that we had in 2016. And to be honest, the chance that we’ll improve the relevant therapies that much in only two years is really pretty slim. In fact, the likely amount of progress in just two years is so small that it might as well be considered zero. Thus, our murine heroes will indeed die in 2019 (or 2020 at best), despite our best efforts.
But now, suppose we develop Robust Human Rejuvenation in 2031, and we take a few dozen 60-year-old humans and duly double their 30-year remaining lifespans. By the time they come back in (say) 2051, biologically 60 again but chronologically 80, they’ll need better therapies, just as the mice did in 2018. But luckily for them, we’ll have had not two but twenty years to improve the therapies. And 20 years is a very respectable period of time in technology—long enough, in fact, that we will with very high probability have succeeded in developing sufficient improvements to the 2031 therapies so that those 80-year-olds can indeed be restored from biologically 60 to biologically 40, or even a little younger, despite their enrichment (relative to 2031) in harder-to-repair types of damage. So unlike the mice, these humans will have just as many years (20 or more) of youth before they need third-generation treatments as they did before the second. And so on…
Longevity Escape Velocity
The key conclusion of the logic I’ve set out above is that there is a threshold rate of biomedical progress that will allow us to stave off aging indefinitely, and that that rate is implausible for mice but entirely plausible for humans. If we can make rejuvenation therapies work well enough to give us time to make then work better, that will give us enough additional time to make them work better still, which will … you get the idea. This will allow us to escape age-related decline indefinitely, however old we become in purely chronological terms. I think the term “longevity escape velocity” (LEV) sums that up pretty well.
One feature of LEV that’s worth pointing out is that we can accumulate lead-time. What I mean is that if we have a period in which we improve the therapies faster than we need to, that will allow us to have a subsequent period in which we don’t improve them so fast. It’s only the average rate of improvement, starting from the arrival of the first therapies that give us just 20 or 30 extra years, that needs to stay above the LEV threshold.
In case you’re having trouble assimilating all this, let me describe it in terms of the physical state of the body. Throughout this book, I’ve been discussing aging as the accumulation of molecular and cellular “damage” of various types, and I’ve highlighted the fact that a modest quantity of damage is no problem—metabolism just works around it, in the same way that a household only needs to put out the garbage once a week, not every hour. In those terms, the attainment and maintenance of escape velocity simply means that our best therapies must improve fast enough to outweigh the progressive shift in the composition of our aging damage to more repair-resistant forms, as the forms that are easier to repair are progressively eliminated by our therapies. If we can do this, the total amount of damage in each category can be kept permanently below the level that initiates functional decline.
Another, perhaps simpler, way of looking at this is to consider the analogy with literal escape velocity, i.e. the overcoming of gravity. Suppose you’re at the top of a cliff and you jump off. Your remaining life expectancy is short—and it gets shorter as you descend to the rocks below. This is exactly the same as with aging: the older you get, the less remaining time you can expect to live. The situation with the periodic arrival of ever better rejuvenation therapies is then a bit like jumping off a cliff with a jet-pack on your back. Initially the jetpack is turned off, but as you fall, you turn it on and it gives you a boost, slowing your fall. As you fall further, you turn up the power on the jetpack, and eventually you start to pull out of the dive and even start shooting upwards. And the further up you go, the easier it is to go even further.
The political and social significance of discussing LEV
I’ve had a fairly difficult time convincing my colleagues in biogerontology of the feasibility of the various SENS components, but in general I’ve been successful once I’ve been given enough time to go through the details. When it comes to LEV, on the other hand, the reception to my proposals can best be described as blank incomprehension. This is not too surprising, in hindsight, because the LEV concept is even further distant from the sort of scientific thinking that my colleagues normally do than my other ideas are: it’s not only an area of science that’s distant from mainstream gerontology, it’s not even science at all in the strict sense, but rather the history of technology. But I regard that as no excuse. The fact is, the history of technology is evidence, just like any other evidence, and scientists have no right to ignore it.
Another big reason for my colleagues’ resistance to the LEV concept is, of course, that if I’m seen to be right that achievement of LEV is foreseeable, they can no longer go around saying that they’re working on postponing aging by a decade or two but no more. There is an intense fear within the senior gerontology community of being seen as having anything to do with radical life extension, with all the uncertainties that it will surely herald. They want nothing to do with such talk. You might think that my reaction to this would be to focus on the short term: to avoid antagonising my colleagues with the LEV concept and its implications of four-digit lifespans, in favour of increased emphasis on the fine details of getting the SENS strands to work in a first-generation form. But this is not an option for me, for one very simple and incontrovertible reason: I’m in this business to save lives. In order to maximise the number of lives saved—healthy years added to people’s lives, if you’d prefer a more precise measure—I need to address the whole picture. And that means ensuring that the general public appreciate the importance of this work enough to motivate its funding.
Now, your first thought may be: hang on, if indefinite life extension is so unpalatable, wouldn’t funding be attracted more easily by keeping quiet about it? Well, no—and for a pretty good reason. The world’s richest man, Bill Gates, set up a foundation a few years ago whose primary mission is to address health issues in the developing world. This is a massively valuable humanitarian effort, which I wholeheartedly support, even though it doesn’t directly help SENS at all. I’m not the only person who supports it, either: in 2006 the world’s second richest man, Warren Buffett, committed a large proportion of his fortune to be donated in annual increments to the Gates Foundation.
The eagerness of extremely wealthy individuals to contribute to world health is, in more general terms, an enormous boost for SENS. This is mainly because a rising tide raises all boats: once it has become acceptable (even meritorious) among that community to be seen as a large-scale health philanthropist, those with “only” a billion or two to their name will be keener to join the trend than if it is seen as a crazy way to spend your hard-earned money. But there’s a catch. That logic only works if the moral status of SENS is seen to compare with that of the efforts that are now being funded so well. And that’s where LEV makes all the difference. SENS therapies will be expensive to develop and expensive to administer, at least at first. Let’s consider how the prospect of spending all that money might be received if the ultimate benefit would be only to add a couple of decades to the lives of people who are already living longer than most in the developing world, after which those people would suffer the same duration of functional decline that they do now.
It’s not exactly the world’s most morally imperative action, is it?
Indeed, I would go so far as to say that, if I were in control of a few billion dollars, I would be quite hesitant to spend it on such a marginal improvement in the overall quality and quantity of life of those who are already doing better in that respect than most, when the alternative exists of making a similar or greater improvement to the quality and quantity of life of the world’s less fortunate inhabitants.
The LEV concept doesn’t make much difference in the short term to who would benefit from these therapies, of course: it will necessarily be those who currently die of aging, so in the first instance it will predominantly be those in wealthy nations. But there is a very widespread appreciation in the industrialised world—an appreciation that, I feel, extends to the wealthy sectors of society—that progress in the long term relies on aiming high, and in particular that the moral imperative to help those at the rear of the field to catch up is balanced by the moral imperative to maximise the average rate of progress across the whole population, which initially means helping those who are already ahead. The fact that SENS is likely to lead to LEV means that developing SENS gives a huge boost to the quality and quantity of life of whomever receives it: so huge, in fact, that there is no problem justifying it in comparison the alternative uses to which a similar sum of money might be put. The fact that lifespan is extended indefinitely rather than by only a couple of decades is only part of the difference that LEV makes, of course: arguably an even more important difference in terms of the benefit that SENS gives is that the whole of that life will be youthful, right up until a beneficiary mistimes the speed of an oncoming truck. The average quality of life, therefore, will rise much more than if all that was in prospect were a shift from (say) 7:1 to 9:1 in the ratio of healthy life to frail life.
Quantifying longevity escape velocity more precisely
I hope I have closed down the remaining escape routes that might still have remained for those still seeking ways to defend a rejection of the SENS agenda. I have shown that SENS can be functionally equivalent to a way to eliminate aging completely, even though in actual therapeutic terms it will only be able to postpone aging by a finite amount at any given moment in time. I’ve also shown that this makes it morally just as desirable—imperative, even—as the many efforts into which a large amount of private philanthropic funding is already being injected.
I’m not complacent though: I know that people are quite ingenious when it comes to finding ways to avoid combating aging. Thus, in order to keep a few steps ahead, I have recently embarked on a collaboration with a stupendous programmer and futurist named Chris Phoenix, in which we are determining the precise degree of healthy life extension that one can expect from a given rate of progress in improving the SENS therapies. This is leading to a series of publications highlighting a variety of scenarios, but the short answer is that no wool has been pulled over your eyes above: the rate of progress we need to achieve starts out at roughly a doubling of the efficacy of the SENS therapies every 40 years and actually declines thereafter. By “doubling of efficacy” I mean a halving of the amount of damage that still cannot be repaired.
So there you have it. We will almost certainly take centuries to reach the level of control over aging that we have over the aging of vintage cars—totally comprehensive, indefinite maintenance of full function—but because longevity escape velocity is not very fast, we will probably achieve something functionally equivalent within only a few decades from now, at the point where we have therapies giving middle-aged people 30 extra years of youthful life. I think we can call that the fountain of youth, don’t you?”
SENS (Strategies for Engineered Negligible Senescence)
http://sens.org/
http://methuselahfoundation.org/
http://mfoundation.org/
http://us.cnn.com/video/?/video/international/2009/11/30/vs.clinic.immortality.cnn
http://longevitymeme.org/topics/strategies_for_engineered_negligible_senescence.cfm
DONATE or COMPETE
https://mfoundation.org/index.php?pagename=mj_donations_donate
http://mfoundation.org/?pagename=mj_mprize_how
http://mfoundation.org/index.php?pagename=mj_mprize_overview
“The Mprize competition is an exciting and viable mid-term strategy to deliver on the Methuselah Foundation’s mission of extending healthy human life. It directly accelerates the development of revolutionary new life extension therapies by awarding two cash prizes: one to the research team that breaks the world record for the oldest-ever mouse; and one to the team that develops the most successful late-onset rejuvenation. Previous winners have already proven that healthy life can be extended; each new winner pushes the outer limits of healthy life back even further…and each new winner takes us even further.
Why Mice?
Because of the mouse’s availability, size, low cost, ease of handling, and strong genetic similarity to humans, there is no other experimental animal that offers such a wide variety of uses to science and medicine. Mice are widely considered to be the prime model of inherited human disease and studies have shown that mice share 99% of their genes with humans. The similarities between sections of human and mouse DNA allow researchers working with mouse genes to make incredibly accurate predictions about the location and function of their human counterparts. To this point, mice have been the mainstay of laboratory research on human illness, and they will continue to be essential players in future studies.”

NAKED MOLE RATS
http://longevitymeme.org/news/view_news_item.cfm?news_id=2774
http://fightaging.org/archives/2008/01/naked-molerats-and-negligible-senescence.php
“Naked mole-rats live in captivity for more than 28.3 years, approximately 9 times longer than similar-sized mice. They maintain body composition from 2 to 24 years, and show only slight age-related changes in all physiological and morphological characteristics studied to date. Surprisingly breeding females show no decline in fertility even when well into their third decade of life. Moreover, these animals have never been observed to develop any spontaneous neoplasm. As such they do not show the typical age-associated acceleration in mortality risk that characterizes every other known mammalian species and may therefore be the first reported mammal showing negligible senescence over the majority of their long lifespan. Clearly physiological and biochemical processes in this species have evolved to dramatically extend healthy lifespan. The challenge that lies ahead is to understand what these mechanisms are.”
AGELESS ANIMALS
http://agelessanimals.org/

NEGLIGIBLE SENESCENCE
http://smart-publications.com/articles/MOM-guerin.php
Learning From Ageless Animals: An Interview with John Guerin
by David Jay Brown
“John Guerin is the founder and director of the AgelessAnimals Project—also known as the Centenarian Species and Rockfish Project. This long-range research project involves investigators at fourteen universities around the world who study animals that don’t seem to age.
There are certain species of rockfish, whales, turtles, and other animals that are known to live for hundreds of years without showing any signs of aging—a phenomenon known to biogerontologists as “negligible senescence.” No one knows for sure how long these animals can live for, but we know that they can live for over two hundred years without showing any observed increase in mortality or any decrease in reproductive capacity due to age. Striking examples are a 109 year old female rockfish that was captured in the wild while swimming around with fertilized eggs, and a hundred-plus year old male whale that was harpooned while it was having sex. The purpose of the AgelessAnimals Project is to understand why these animals don’t seem to age and then to apply that understanding to human longevity.
Guerin is an experienced project manager, who conceived of the AgelessAnimals project and orchestrates all of the studies. The two principal advisors to this project are Dr. Leonard Hayflick and Dr. Aubrey de Grey, both of whom were also interviewed for the Mavericks of Medicine collection. Dr. Hayflick, discoverer of the “Hayflick limit” of cellular senescence, states that “Guerin’s project is not only unique, but probes an area of almost total neglect in biogerontology, yet an area with more promise to deliver valuable data than, perhaps, any other.”
When I asked Dr. de Grey about the importance of studying ageless animals he said, “All organisms with organs that rely on the indefinite survival of individual non-dividing cells (such as neurons in the brain) should age, though some, including humans, age very slowly. Some species do even better—we cannot yet measure their rate of aging at all—and studying them may well reveal ways to slow our own aging.”
In addition to coordinating and orchestrating the AgelessAnimals project, Guerin lectures regularly on the subject of ageless animals. To find out more about Guerin’s work and the AgelessAnimals Project visit their Web site: www. agelessanimals.org. John Guerin seemed eager and excited to discuss his project with me. We spoke about some of the latest research that’s going on with long-lived animals, why this type of research has been neglected for so long, and how studying ageless animals might help us to understand the aging process better and extend the human lifespan.
Q: What inspired the AgelessAnimals Project?
John Guerin: Back in 1995, I began looking into biotech, biogerontology, and the studies of aging. I read many different books, articles, and scientific papers. The turning point came when I read Dr. Leonard Hayflick’s book How and Why We Age. Dr. Hayflick had a chapter called “Some Animals Age, Some Do Not,” and I thought, wow, now that’s interesting. I’d heard rumors and old wive’s tales about how some animals live for an extraordinarily long time, but this was the first time that I had come across that information from a scientific source. So I started doing some research on long-lived animals, and I found out that there’s very little known. On my web site, I have some references available.
I met Dr. Hayflick at a Gerontological Society of America meeting in November of 1995, and I told him about my project management background. I said I’d like to join whoever is working in this area, and I asked him who is. His answer was, “Nobody is, but they should be.” So I tried to get something going on my own. I did a lot of research on different animals. I spent about a year looking at koi—the fancy Japanese carp—and it’s very likely that they do live quite a long time, at least over fifty years. They were reputed to live over two hundred years, but the readings were based on scales, and those are not accurate. So they didn’t turn out to be a good candidate to study.
Then in 1997, I got some data from the Alaska Fish and Game. There’s a chart at the bottom of my Web page with a rockfish on it that shows ages for different rockfish that were caught off the coast of Alaska, and the range is between twelve and 107 years. Now, that’s a randomly caught sampling—it wasn’t like they were trying to get older individuals.
Those were the ones that fishermen caught and were going to people’s dinner tables that evening. So when I realized that individuals at those ages were available, I became very interested. We got samples from the Alaska Fish and Game in 1997. I say “we” because by then I had a couple of researchers at Oregon State University and the Linus Pauling Institute interested in looking at the rockfish. So the Alaska Fish and Game sent us five older rockfish. After we aged them, it turned out that the the youngest rockfish that they sent us was 79 years old, and the oldest was a 109 year old female that still had eggs.
Q: Isn’t that extraordinary?
John Guerin: Yeah, and kind of sad. It’s like, how long would this fish have lived if it wasn’t caught? It didn’t die of old age. It was fertile and still going strong in the ocean at 109 when they caught it. So that helped us to focus the project on rockfish. We have had one study on turtles. Whales are a very fascinating subject too, because they’re warm-blooded mammals like we are, and they’ve now been documented to be over two hundred years of age.
Q: What is the goal of the AgelessAnimals Project?
John Guerin: Quite simply, the goal is to understand the genetic and biochemical processes that long-lived animals use to retard aging. These long-lived animals have what’s technically called “negligible senescence,” as defined by Caleb Finch at the University of Southern California in Longevity, Senescence, and the Genome (1995).
Q: What is negligible senescence?
John Guerin: Basically, this refers to an animal species that doesn’t show any significant signs of aging as it grows older. Unlike humans and most other mammals, there’s no decrease in reproduction after maturity.
There’s also no notable increase in mortality rate with age, but that’s a little harder to prove. I’ve been talking with a statistician and he’s asking, how do you know? To do a study of this type would take a couple of hundred years to complete. But compared to us there’s no noted increase in mortality rate. I mean, if you are ninety years old, you’re much more likely to die next year then you are if you’re only twenty years old. But we don’t seem to see any increase in mortality with rockfish and several of these other animals over time.
Q: Why do you think these animals can live for so long without showing any signs of aging?
John Guerin: The purpose of the project is to understand why, and how to apply it to extending the health and lifespan of humans. My background is in business project management; I have a project management professional certification. I’m not a bioresearcher, a biochemist, or a biogerontologist—but I’m the one who organizes it all, and gets everyone involved. I get the researchers the samples and all that.
Actually, I thought I had a better idea about why these animals have negligible senescence when I started this project ten years ago. But it’s hard to say. Back then we didn’t know whales lived that long. That whales can live for over two hundred years was just discovered in the last five years. Up until then we thought that humans lived longer than any other mammal. So why certain animals would live much longer than others, and much longer than we do as a matter of fact—pretty much double what we’ve known humans to live—we don’t understand.
There are some people who think that this can’t be so, that this would violate the evolutionary theory of senescence, because nature doesn’t select for longevity. But that’s not necessarily true, because what’s commonly seen is that there’s just such a high mortality rate in nature.
Even for humans, probably before two thousand years ago, we didn’t live very long. We were hunted by tigers and wild animals, and traits of longevity, presumably, weren’t selected for. But if these animals, like the rockfish, can be 109 years old and still be reproductive, nature is going to allow those genes to keep contributing to the gene pool, so that it won’t select against longevity.
Q: So we don’t know if these animals are simply aging more slowly or not at all? Since we have haven’t found any rockfish or whales that live for three or four hundred years, that might suggest that there is a certain limit on how long they can live.
John Guerin: Well, we just do not know. We honestly do not know. It really is unfortunate that there is so little known in this field. Ecologists have never thought of this in the terms that gerontologists are now thinking of it in. To give you an idea, let’s say you have a sample of a species, and you see they live to twenty years. That’s the oldest you sample out of several hundred. Then that’s their maximum longevity. That’s really the basis of their thinking in most cases. Mice, as you probably well know, don’t live for more than a couple years, even in the best laboratory environment, with all the best nutrients and all the best food. They don’t live very long. They just can’t. They’ll start having all sorts of age-related pathological functions, and they’ll die of old age.
But this other group of organisms, those that possess what Finch termed “negligible senescence,” they don’t seem to be showing the classical signs of aging that we’re used to. So, who is to say the longest they could live? As an example, in Finch’s book that was published in 1990, at that time the longest lived whale was—I believe it was a Blue Whale—something like 108 years old. That’s like, okay, well that’s not so startling. Humans live longer than that. We’re mammals. They’re mammals. We live longer. Then a study was done on bowhead whales, and they found that out of forty whales sampled, four of them were over a hundred years old, and one of them was over two hundred years old. And they didn’t die of old age either—they were harpooned. I have a reference on the web site.
Q: How might studying ageless animals help us to understand human aging better?
John Guerin: By understanding how other animals are naturally able to live a lot longer than we are, we can ask: What is genetically different? What is biochemically different? There are two major problems with studying long-lived animals. One is that nobody knows what causes aging. If you’re able to say what causes aging, that’d be really easy then to target that same factor in animals that are living a very long time—whales, rockfish, sturgeon, lobsters, and several other animals—and then you could study it. If you looked on our web site you’ve seen that we did studies in everything from lisosones to microarrays to telomere-telomerase activity, because you just don’t know. That’s one problem. The other is that these animals live so long that you have to ask: How do you do an experiment? Let’s say we think a certain gene’s involved in aging, so we were going to do a knockout. Then instead of living two hundred years a rockfish lives seventy-five years.
Q: Wouldn’t it take quite awhile to run the experiment?
John Guerin: Yeah, it would be somebody else finishing it up, and it certainly wouldn’t be of much practical benefit. So the direction we’re taking in the project is we’re looking at the difference between long-lived rockfish and short-lived rockfish. The other thing is basically identifying genetic differences, and going at it that way, because there’s no practical way you could run an experiment that would go on for decades.
At first rockfish just seemed to be a good model, or a handily available model. They’re commercially caught, go on the dinner table, and we were able to get lots of samples of them. Then, of course, the news about whales came out, which is very intriguing, and there are lots of other animals that are either known or suspected to live a very long time. But the really intriguing thing about rockfish is that in the same genus—which is sebastes—there are rockfish that have not been noted more than about twelve to twenty years maximum longevity, and these are essentially cousins. They are rockfish, and some of these at least have been caught in thousands of samples, so it’s not just an aberration of a small sample size.
One of the key issues people have raised to me at meetings is that you have to have something to compare these long-lived animals to in order to try and understand why they’re successfully retarding aging. So what better model can you have than another species within the same genus that don’t live a very long time? In all the meetings I’ve gone to I haven’t had anybody come up and say, oh here’s another species that has a really diverse longevity. There is one kind of similar one—the naked mole rat. It’s just in the last few years that it’s gotten more publicity. It’s a rodent, and most rodents—like mice and rats—live maybe two to five years maximum. The naked mole rat has been documented to live at least into its twenties. So it’s on quite another order of magnitude different than other rodents. The bat is another exception that lives way longer than other mammals, and birds, of course, are their own interesting exception.
So that’s our focus, which is almost out of necessity, because how do you design an experiment to test for longevity when you’ve got such long-lived animals? Whatever tests we’re going to do to the long-lived rockfish in the future, like a micro-array, we want to do with the short-lived ones too.
Q: Could you talk about some of the principal investigators for the Ageless Animal project, and can you summarize some of the latest research that’s going on with long-lived animals?
John Guerin: There’s fourteen principal investigators at fourteen different universities. There are some co-PIs involved as well, such as Dr. Judd Aiken at the University of Wisconsin, Madison. He’s very well-known and respected in the field. He does mitochondrial mutation studies, and this could be one of the more important areas because of what we know about free radical damage. The oxidative theory seems to be of the more important theories of aging, so I’m encouraged, even though at this point he hasn’t gotten results yet. His lab is working on amplifying the primers. So that could be a very important study. I think the microarray study is an important one too.
Dr. Ana Maria Cuervo, who’s at Albert Einstein College of Medicine in New York City, did the most complete study. Her study was on lisosomes and proteolytic activity, and she actually has done more than is on the web site. She added some more tests that she didn’t have available a couple of years ago, and she told me about a month ago that she was getting her manuscript ready to publish. So that would be the most extensive study. Also, there’s Glenn Gerhard, M.D., who was at Dartmouth and then he took a full-time research position at a research institute in Pennsylvania. He did a SOD (superoxide dismutase) study and then also the microarray study.
Q: Why do you think that the study of long-lived animals has been neglected for so long?
John: I have thought about it, and partly I have to say I don’t understand why. I think that’s why somebody like me would get involved, because I have a project management background and I can see the big picture. There’s more than one reason I can see as to why people in the field wouldn’t have gotten involved. It’s risky to put your career on the line to look at animals that haven’t been studied very well and that there’s no cultures available of. Whereas with other species strains are easily available. For instance, with any mouse you want you could get a strain, and you could have it under the identical conditions you need.
But this hasn’t been done with any of these long-lived animals. For me, the biggest question really is: Why hasn’t the National Institute of Aging taken a lead? This is a perfect opportunity for government to get involved, where there is no profit motive. This is basic research that we’re doing with these animals, and basic research doesn’t necessarily have a pay-back. Now, let’s say we find something like we did with the SOD study. We had a very interesting finding that SOD went up with age in rockfish, and as you may be aware, SOD is the strongest antioxidant in our bodies, and in most animals. So that it goes up with age is a very intriguing finding. That’s something we hope to look into more, but in general all of the things we’ve done are just basic science. We’re just laying the groundwork.
Q: Has anybody done any studies to see if whale cells, rockfish cells, or turtle cells reach a Hayflick limit in the number of times that they can divide? Are their telomeres growing shorter with each cell division?
John Guerin: In terms of the Hayflick limit, you very well may be aware that most gerontologists don’t consider that to be a limitation of aging. At some point, maybe about ten years ago, it was a much bigger topic. Nowadays, telomeres and telomerase is much more of a cancer issue, because most cancer cells keep producing that enzyme that allows cells to keep dividing.
Q: When I interviewed Dr. Michael Fossel for this collection he thought differently.
John Guerin: I would have to say that the majority of gerontologists don’t believe it that way. I remember at a meeting a couple of years ago, somebody just making an offhand remark that we used to think telomerase and telomeres were important. I think if you do a survey you would find that that the majority of gerontologists don’t believe it that way. The telomerase limit and the Hayflick limit don’t seem to necessarily be what it once was thought to be, because even older people have continued replication of the cells that do divide. There’s a bunch of reasons that it doesn’t seem as important now as at one point when they thought it was.
Q: Yes, I understand that, but I’m still curious. Do you know if these animals that have negligible senescence, if their cells reach a Hayflick limit? Is there a limit to how many times the cells from these animals can divide?
John Guerin: We have fourteen studies—twelve in the U.S. and two in Europe. One of the European studies is in Germany by someone named Guido Krupp, who looked at telomerase levels in nine different rockfish. He looked at three samples—one of heart, one of liver, and one of brain—all the way from teenage years up to a 93 year old rockfish. All of the three tissues showed expression of telomerase, and there was no age-dependent change of expression of telomerase in the tissues. There was individual differences. Some were higher and some were lower. One of the higher levels of telomerase was found in the 93 year old, but the primary finding was that there was no trend. As far as whales go, the only other person I know outside our group and Caleb Finch at USC, who actually is studying these long-lived animals is a guy at the University of Texas in Dallas named Jerry Shay. He got some samples of bowhead whale, and he’s basically doing these cell replications to see how many replications he gets out of them.
Apparently, it’s pretty hard to get the samples, because they had to go through the Canadian government, and it was quite an ordeal. Jerry Shay is the only one I know of who’s done bowhead whale studies. But in this one study four of the whales out of forty were documented to be over a hundred years old, and one of them was over two hundred years old. And that’s without doctors. Although this was not in the paper, we know that at least one of those hundred-plus year old male whales was reproductive, because when he was harpooned he was caught in the act.
Q: Wait a minute. This hundred-plus year old whale was harpooned while it was having sex?
John Guerin: Yes, and it was over a hundred. There were three that were between a hundred and two hundred. One was 211 years old. When I talked to the researcher, who is an ecologist, I said, “Gerontologists want to know, how do you know that the whales weren’t about to keel over, that they were on their last leg?” And then he has an example like this. I’m like, were they reproductive? And he goes, well, one of the males sure was.”

LOBSTERS IMMORTAL?
http://npr.org/templates/story/story.php?storyId=11382976
http://animals.howstuffworks.com/marine-life/400-pound-lobster.htm/
Big Lobsters and Eternal Life
“Decline is an accepted part of old age for most people, even for those still searching for the fountain of youth. We expect the same in our pets and in the flies that buzz around us, albeit at a different rate. So why are lobsters different? A study conducted in 1998 showed that lobsters maintain telomerase activation late in life. But before we explain that, let’s talk briefly about cell division.
Telomeres are like caps or sheathes that encase the ends of chromosomes. When cells divide, telomeres get shorter. When telomeres get to a certain length, they can no longer protect chromosomes and the chromosomes start to suffer damage. The number of cell divisions before damage sets in is called the Hayflick limit. Telomerase is an enzyme that adds length to telomeres, extending their life span. In humans, telomerase is abundant in embryonic stem cells and then declines later in life. This is actually a good thing because when cells re-activate telomerase after reaching the Hayflick limit, they become cancerous (in other words, they don’t die when they’re supposed to). The downside is that cells with short telomeres weaken and die, so we eventually die, too.
In humans, telomerase levels decline later in life and are only found in some types of tissue, but in lobsters, telomerase is found in all types of tissue. That likely accounts for lobsters’ ability to grow throughout their lives. And because lobsters’ skeletons are on the outside and the molting process allows them to periodically shed their exoskeletons in favor of a new, larger one, their constant growth isn’t a problem. With a steady, evenly distributed supply of telomerase, lobsters don’t approach the Hayflick limit, which means that their cells stay pristine, young and dividing.
The dual role of telomerase in keeping cells healthy and in cancer growth means that it’s an important area of research for both anti-aging and cancer treatments. Further study of lobsters may teach us more about their longevity, how long they can actually live and what that knowledge may mean for human health. Scientists are also studying a variety of other animals that are long-lived. Like lobsters, many types of turtles don’t show compromised immunity or physical breakdown because of age. They also become more fertile with age and usually die because of a predator or malady unrelated to age.
A bird known as Leach’s storm petrel fits into a human hand yet lives more than 30 years. They’re also the only known animal in which telomeres grow longer with age. Related animal species with vastly different life spans are also a point of interest. Conventional mice live only three years, but naked mole rats can live for 28. Other animals being studied include whales, bats, rockfish, zebrafish and clams, the oldest of which, a quahog clam, lived to be 220 years old. In many of these animals, the rate of telomere deterioration corresponds with their lifespan. The longer the telomeres last, the longer the animals live. Studying these creatures may tell us much about human aging and lead to treatments for aging-related diseases. Exciting research is being conducted on many fronts — on the molecular and genetic levels and regarding lifestyle, diet and habitat. If one day humans discover an important new treatment for cancer, it may be due to one of these creatures — or to a 200-pound lobster living peacefully in a tank at Boston University.”
CONTACT
Jelle Atema
http://people.bu.edu/atema/
http://bu.edu/biology/people/faculty/atema/
email : atema [at] bu [dot] edu
FOREVER
http://kuro5hin.org/story/2004/12/20/184723/82
http://metafilter.com/88971/Nobody-Home
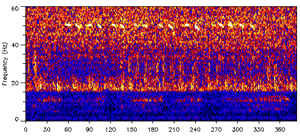
“For the past 21 years, across the limitless expanse of the North Pacific, a lonely whale has been singing, calling for a response. There has been none, and there never will. Picked up first in 1989 by NOAA hydrophones, the call is clearly a whale, but different than all other known species. Different enough that no other whale has responded in all this time. Hypotheses vary, but the mental image is definitely haunting.”
ALONE at 52 HERTZ
http://pmel.noaa.gov/vents/acoustics/whales/sounds/whalewav/ak52_10x.wav
http://pmel.noaa.gov/vents/acoustics/whales/sounds/sounds_52blue.html
http://newscientist.com/article/dn6764-lonely-whales-song-remains-a-mystery.html
Lonely whale’s song remains a mystery
by Jon Copley / 10 December 2004
“Marine biologist Mary Ann Daher of Woods Hole Oceanographic Institution in Massachusetts, US, and her colleagues used signals recorded by the US navy’s submarine-tracking hydrophones to trace the movements of whales in the north Pacific. The partially declassified records show that a lone whale singing at around 52 hertz has cruised the ocean every autumn and winter since 1992. Its calls do not match those of any known species, although they are clearly those of a baleen whale, a group that includes blue, fin and humpback whales.
Blue whales typically call at frequencies between 15 and 20 hertz. They use some higher frequencies, but not 52 hertz, Daher says. Fin whales make pulsed sounds at around 20 hertz, while humpbacks sing at much higher frequencies. The tracks of the lone whale do not match the migration patterns of any other species, either. Over the years the calls have deepened slightly, perhaps because the whale has aged, but its voice is still recognisable. Daher doubts that the whale belongs to a new species, although no similar call has been found anywhere else, despite careful monitoring.”
CONTACT
Mary Ann Daher
http://whoi.edu/profile/mdaher/
email : mdaher [at] whoi [dot] edu


