BANANA HALFLIFE
http://physics.isu.edu/radinf/natural.htm
https://chemtable.wordpress.com/2014/04/05/banana-equivalent-dose-or-bed/
Banana Equivalent Dose — or BED / April 5, 2014
“Many worry about radiation exposure — particularly after Japan’s 2011 Fukushima Daiichi nuclear plant disaster released significant amounts of radionuclides, such as iodine-131 and cesium-137 into the atmosphere and ocean. Increased exposure to radiation may also come form living near a nuclear power plant, flying in an airplane, or from undergoing certain medical procedures, such as X-rays, mammograms or CT scans. It’s important to remember that radiation is a natural factor in the environment. You are exposed to background radiation in the air, in the soil, in the food we eat, even in our own bodies. In your home, radiation comes from typical construction materials, such as bricks and wallboard, granite countertops and tile floors, through cat litter, or even from the soil or bedrock beneath your house. On average, 82% of your total annual exposure to radiation is from natural sources, most of that from radon. A wonderful graphic (shown above) is from the xkcd website. Note that we’re talking about ionizing radiation, the type of exposure absorbed by body tissue that can potentially damage human cells.
 One measure that has been proposed to illustrate natural radiation exposure is the Banana Equivalent Dose (more technically the biologically effective dose) or BED. This refers to the radiation exposure from eating one banana. In fact, all foods are slightly radioactive, but bananas are particularly rich in potassium, which is the major source of natural radioactivity in plant matter. And 0.1% of potassium consists of the radioactive isotope Potassium-40 (40K), which decays with a half-life of 1.25 billion years. For an average banana, that yields about 14 decays per second. This translates into a microscopic amount of radiation, which is quantified as 0.1 micro Sievert or mSv.
One measure that has been proposed to illustrate natural radiation exposure is the Banana Equivalent Dose (more technically the biologically effective dose) or BED. This refers to the radiation exposure from eating one banana. In fact, all foods are slightly radioactive, but bananas are particularly rich in potassium, which is the major source of natural radioactivity in plant matter. And 0.1% of potassium consists of the radioactive isotope Potassium-40 (40K), which decays with a half-life of 1.25 billion years. For an average banana, that yields about 14 decays per second. This translates into a microscopic amount of radiation, which is quantified as 0.1 micro Sievert or mSv.
Obviously bananas won’t kill you. They won’t set off a Geiger counter; they don’t glow in the dark. There are rumors that crates of bananas have set off radiation detectors at customs — but these are most likely urban legends. Trace levels of radiation are also found in foods such as beans, nuts, seeds and potatoes. Even human bodies are naturally radioactive. Sleeping next to someone for eight hours yields a dose of 0.05 mSv. This works out to half a BED – which seems quite appropriate! To put it in perspective, fifty bananas would be the equivalent of the radiation dose of a dental X-ray; eating 70,000 bananas would equal one chest CT scan. The yearly dose per person from food is estimated at 400 micro sieverts . The maximum yearly dose permitted for U.S. radiation workers is 50,000 mSv. Is the BED a valid measure of radiation exposure? Maggie Koerth Baker writes that the potassium-40 in bananas is a poor choice, “because the potassium content of our bodies seems to be under homeostatic control. When you eat a banana, your body’s level of Potassium-40 doesn’t increase. You just get rid of some excess Potassium-40. The net dose of a banana is zero.” But it takes time for the body to remove excess potassium, during which time doses can accumulate.”
 A worker using a Geiger counter checks for possible radioactive contamination at Noryangjin Fisheries Wholesale Market in Seoul, South Korea, September 6, 2013.
A worker using a Geiger counter checks for possible radioactive contamination at Noryangjin Fisheries Wholesale Market in Seoul, South Korea, September 6, 2013.
POTASSIUM-40
https://www.americanprogress.org/issues/green/news/2013/09/11/73914/fukushima-fallout-not-affecting-u-s-caught-fish/
http://nextbigfuture.com/2011/03/banana-dose-equivalents-of-radiation.html
http://boingboing.net/2010/08/27/bananas-are-radioact.html
Yes bananas are radioactive — But not a good way to explain radiation exposure
by Maggie Koerth-Baker / Aug 27, 2010
“Just look at that radioactive banana. There’s nothing special about it or where it was grown. All bananas are radioactive, because all bananas contain the radioactive isotope Potassium-40. In fact, a lot of things you might not suspect of being radioactive are, including Brazil nuts, and your own body. And this fact is sometimes used to downplay the impact of exposure to radiation via medical treatments or accidental intake. A post by nexusheli on the Submitterator turned me on to the idea of the Banana Equivalent Dose—a way of putting radiation exposure into context by comparing intake from, say, local milk just after the near-disaster at Three Mile Island, to intake from a normal, workaday banana. Wikipedia explains the point of this exercise:
The banana equivalent dose is the radiation exposure received by eating a single banana. Radiation leaks from nuclear plants are often measured in extraordinarily small units (the picocurie, a millionth of a millionth of a curie, is typical). By comparing the exposure from these events to a banana equivalent dose, a more realistic assessment of the actual risk can sometimes be obtained.
This isn’t just about pro-nuclear propaganda. “Picocurie” is one of those words that really doesn’t mean anything to lay people. Confusing units of measurement, when combined with the word “nuclear” can, understandably, freak people out. And, depending on the situation, there isn’t always cause for said freak out. Having a way of explaining what picocurie means, in the context of everyday, normal, safe exposures, really is a useful tool for putting radioactivity into a context the public can understand. But, the Banana Equivalent Dose probably isn’t the best way to do that.
https://www.youtube.com/watch?v=kfgYRIGjNkA
The problem is that this system implies that all radioisotopes are created equal—That there’s no difference between 520 picocuries of Potassium-40 and a similar intake of, say, radioactive iodine. And that simply isn’t true. I contacted Geoff Meggitt — a retired health physicist, and former editor of the Journal of Radiological Protection—to find out more. Meggitt worked for the United Kingdom Atomic Energy Authority and its later commercial offshoots for 25 years. He says there’s an enormous variation in the risks associated with swallowing the same amount of different radioactive materials—and even some difference between the same dose, of the same material, but in different chemical forms.
It all depends on two factors:
1)The physical characteristics of the radioactivity—i.e, What’s its half-life? Is the radiation emitted alpha, beta or gamma?
2) The way the the radioactivity travels around and is taken up by the body—i.e., How much is absorbed by the blood stream? What tissues does this specific isotope tend to accumulate in?
The Potassium-40 in bananas is a particularly poor model isotope to use, Meggitt says, because the potassium content of our bodies seems to be under homeostatic control. When you eat a banana, your body’s level of Potassium-40 doesn’t increase. You just get rid of some excess Potassium-40. The net dose of a banana is zero. And that’s the difference between a useful educational tool and propaganda. (And I say this as somebody who is emphatically not against nuclear energy.) Bananas aren’t really going to give anyone “a more realistic assessment of actual risk”, they’re just going to further distort the picture.”
[Geoff Meggitt has written a book about the history of radiation and protection, called Taming the Rays.]
https://www.youtube.com/watch?v=RSkVuh9bV-U
JUST BANANAS
http://io9.com/10-of-the-most-absurd-units-of-measurement-on-earth-1694474719
http://www.forbes.com/sites/timworstall/2013/08/10/the-fukushima-radiation-leak-is-equal-to-76-million-bananas/
Fukushima Radiation Leak Is Equal To 76 Million Bananas
by Tim Worstall / Aug 10, 2013
“There’s much screaming and shouting from the usual suspects about the new radiation leak discovered at Fukushima, the stricken nuclear power plants in Japan. What they’re not telling you is that the radiation leakage is around the same as 76 million bananas. A fact which should help to put it all into some perspective. Here’s Greenpeace:
Environmental group Greenpeace said Tepco had “anxiously hid the leaks” and urged Japan to seek international expertise. “Greenpeace calls for the Japanese authorities to do all in their power to solve this situation, and that includes increased transparancy…and getting international expertise in to help find solutions,” Dr Rianne Teule of Greenpeace International said in an e-mailed statement.
Not that Greenpeace is ever going to say anything other than that nuclear power is the work of the very devil of course. And the headlines do indeed seem alarming: “Radioactive Fukushima groundwater rises above barrier – Up to 40 trillion becquerels released into Pacific ocean so far – Storage for radioactive water running out.” Or: “Tepco admitted on Friday that a cumulative 20 trillion to 40 trillion becquerels of radioactive tritium may have leaked into the sea since the disaster.” Most of us haven’t a clue what that means of course. We don’t instinctively understand what a becquerel is in the same way that we do pound, pint or gallons, and certainly trillions of anything sounds hideous. But don’t forget that trillions of picogrammes of dihydrogen monoxide is also the major ingredient in a glass of beer. So what we really want to know is whether 20 trillion becquerels of radiation is actually an important number. To which the answer is no, it isn’t. This is actually around and about (perhaps a little over) the amount of radiation the plant was allowed to dump into the environment before the disaster. Now there are indeed those who insist that any amount of radiation kills us all stone dead while we sleep in our beds but I’m afraid that this is incorrect. We’re all exposed to radiation all the time and we all seem to survive long enough to be killed by something else so radiation isn’t as dangerous as all that.
At which point we can offer a comparison. Something to try and give us a sense of perspective about whether 20 trillion nasties of radiation is something to get all concerned about or not. That comparison being that the radiation leakage from Fukushima appears to be about the same as that from 76 million bananas. Which is a lot of bananas I agree, but again we can put that into some sort of perspective. Let’s start from the beginning with the banana equivalent dose, the BED. Bananas contain potassium, some portion of potassium is always radioactive, thus bananas contain some radioactivity. This gets into the human body as we digest the lovely fruit (OK, bananas are an herb but still…): “Since a typical banana contains about half a gram of potassium, it will have an activity of roughly 15 Bq.” Excellent, we now have a unit that we can grasp, one that the human mind can use to give a sense of proportion to these claims about radioactivity. We know that bananas are good for us on balance, thus this amount of radioactivity isn’t all that much of a burden on us. We also have that claim of 20 trillion becquerels of radiation having been dumped into the Pacific Ocean in the past couple of years. 20 trillion divided by two years by 365 days by 24 hours gives us an hourly rate of 1,141,552,511 becquerels per hour. Divide that by our 15 Bq per banana and we can see that the radiation spillage from Fukushima is running at 76 million bananas per hour. Which is, as I say above, a lot of bananas. But it’s not actually that many bananas. World production of them is some 145 million tonnes a year. There’s a thousand kilos in a tonne, say a banana is 100 grammes (sounds about right, four bananas to the pound, ten to the kilo) or 1.45 trillion bananas a year eaten around the world. Divide again by 365 and 24 to get the hourly consumption rate and we get 165 million bananas consumed per hour.
We can do this slightly differently and say that the 1.45 trillion bananas consumed each year have those 15 Bq giving us around 22 trillion Bq each year. The Fukushima leak is 20 trillion Bq over two years: thus our two calculations agree. The current leak is just under half that exposure that we all get from the global consumption of bananas. Except even that’s overstating it. For the banana consumption does indeed get into our bodies: the Fukushima leak is getting into the Pacific Ocean where it’s obviously far less dangerous. And don’t forget that all that radiation in the bananas ends up in the oceans as well, given that we do in fact urinate it out and no, it’s not something that the sewage treatment plants particularly keep out of the rivers. There are some who are viewing this radiation leak very differently:
Arnold Gundersen, Fairewinds Associates: […] we are contaminating the Pacific Ocean which is extraordinarily serious.
Evgeny Sukhoi: Is there anything that can be done with that, I mean with the ocean?
Gundersen: Frankly, I don’t believe so. I think we will continue to release radioactive material into the ocean for 20 or 30 years at least. They have to pump the water out of the areas surrounding the nuclear reactor. But frankly, this water is the most radioactive water I’ve ever experienced.
I have to admit that I simply don’t agree. I’m not actually arguing that radiation is good for us but I really don’t think that half the radiation of the world’s banana crop being diluted into the Pacific Ocean is all that much to worry about. And why we really shouldn’t worry about it all that much. The radiation that fossil fuel plants spew into the environment each year is around 0.1 EBq. That’s ExaBecquerel, or 10 to the power of 18. Fukushima is pumping out 10 trillion becquerels a year at present. Or 10 TBq, or 10 of 10 to the power of 12. Or, if you prefer, one ten thousandth of the amount that the world’s coal plants are doing. Or even, given that there are only about 2,500 coal plants in the world, Fukushima is, in this disaster, pumping out around one quarter of the radiation that a coal plant does in normal operation. You can worry about it if you want but it’s not something that’s likely to have any real measurable effect on anyone or anything.”
 https://xkcd.com/radiation/sources.html
https://xkcd.com/radiation/sources.html
REFERENCE BANANA
https://en.wikipedia.org/wiki/Banana_equivalent_dose#History
https://web.archive.org/web/20090514034843/http://www.radlab.nl/radsafe/archives/9503/msg00074.html
http://health.phys.iit.edu/archives/2011-March/031395.html
http://health.phys.iit.edu/extended_archive/9503/msg00074.html
Banana Equivalent Dose
by Gary Mansfield / 7 Mar 1995
re: The Radiation Dose from a “Reference Banana.”
“Some time ago (when I almost had time to do such things) I calculated the dose one receives from the average banana. Here’s how it goes:
On page 620 of the CRD Handbook on Rad Measurement and Protection, the concentration of K-40 in a “Reference Banana” is listed as 3520 picocuries per kilogram of banana. For those of us who are stuck in certain unit ruts, this is equivalent to 3.52E-6 microcuries of K-40 per gram of banana. An average “Reference” banana weighs (masses) about 150 grams (I think.) So, the ICRP Reference Banana contains about 5.28E-4 microcuries of probably deadly K-40. Federal Guidance Report #11 lists the ingestion dose (committed effective dose equivalent) for K-40 as 5.02E-9 Sv/Bq or (again, for those of us who are “unit-challenged,” 1.86E-2 rem per microcurie ingested.) Thus, the CEDE from ingestion of a Reference Banana is 5.28E-4 x 1.86E-2 = 9.82E-6 rem or about 0.01 millirem. I have found this “Banana Equivalent Dose” very useful in attempting to explain infinitesmal doses (and corresponding infinitesmal risks) to members of the public. (Interestingly, the anti-nukes just HATE this, and severely critisize us for using such a deceptive concept.)
https://twitter.com/scale_banana/status/1325148433703870469
Would love to go into more detail, but have to get back to our DEADLY Human Radiation Experiments (i.e., eating bananas.) The same table in the CRC Handbook lists 3400 pCi/kg for white potatoes and 4450 pCi/kg for sweet potatoes – so you could carry through the same sort of calculation for Reference Potatoes. Interestingly, raw lima beans come in at 4640 pCi/kg, “dry, sweet” coconut comes in at 6400 pCi/kg, and raw spinach (yum!) comes in at 6500 pCi/kg. Considering the fact that the DOE has officially stated that “there is no safe dose of radiation” my advice to you all is to stop eating immediately.
Oh yes! Almost forgot. Regarding K-40, go into your local grocery store, buy some salt-substitute (there are two common brands, and the one in the white and orange labeled container works best) spread some out on a table and check it out with a GM survey instrument. There it is folks, deadly radioactivity in your grocery store!
Yours for healthful diets . . .
Captain Internal Dosimetry aka Gary Mansfield, LLNL, (mansfield2 [at] llnl.gov)
Disclaimer:
Neither Lawrence Livermore National Laboratory, the University of California, nor the Department of Energy recommends eating bananas.”
BACKGROUND RADIATION
https://www.quora.com/Is-it-true-that-bananas-are-radioactive-and-if-you-eat-too-many-they-can-kill-you
http://physicsbuzz.physicscentral.com/2014/01/how-radioactive-in-bananas-is-room.html
How Radioactive (in Bananas) is the Room You’re Sitting In Right Now? / January 27, 2014
“According to recent research (and some of my own calculations) sitting in your living room for an hour yields roughly the same dose of radiation as eating half a banana. A team in China is working on improving the way scientists measure a room’s radioactivity calculated how much radiation an average room gives off. Everything emits a trace amount of radiation. Bananas, dirt, seagulls, Abe Vigoda and coffee tables are all to some degree a very tiny bit radioactive. It’s because radioactive elements like potassium-40, uranium-238 and thorium-232 are ubiquitous throughout the planet’s surface, and get absorbed into different materials through various natural processes. They’re part of what makes up the Earth’s “background radiation.”
Building materials like brick and concrete are no different. Tiny amounts of the radioactive potassium, uranium and thorium isotopes are inherently mixed in, meaning that even as you read this, the walls are spraying you with gamma rays. To be clear, the amount of radiation is incredibly tiny. It’s not remotely near any amount that’s dangerous and it’s undetectable to all but the most sensitive of detectors. Radiation is essentially energized particles emitted from an atom’s nucleus when it breaks apart. Different elements emit one of a couple of different kinds or radiation. Alpha particles, are essentially helium nuclei, and can be easily blocked by a piece of paper or your skin, but can be extremely dangerous if swallowed. Beta particles are electrons and can penetrate a bit deeper. Gamma radiation is actually electromagnetic radiation, like a super-powerful X-ray. The researchers quantified exactly how how much of the different kinds of radiation someone sitting in the middle of a room is exposed to from its walls, floor and ceiling. It’s a fair question, because on average a person spends nearly 80 percent of their lives at home or in an office.
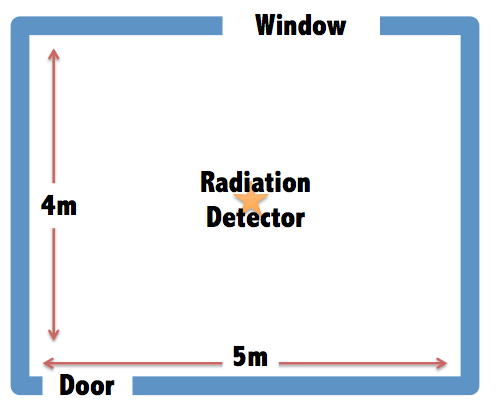
To find out, the researchers designed a virtual room five meters long, four meters wide and 2.8 meters high with one door and one window. The team then calculated how much radiation one would absorb in the middle of that room depending on what the walls were made of and how thick they were. The different materials, brick, marble, granite and four different types of concrete, all have distinct densities and concentrations of radioactive elements. The researchers then plugged in the different dimensions into a special computer program called MCNPX which is used to calculate how radiation interacts with, well, everything. So the scientists input in the room’s dimensions, the various building materials and the half-lives of the different isotopes mixed in to figure out how much radiation is emitted by the building itself and how much of that a person absorbs at any point in the room. The unit used to measure how much radiation a person absorbs is the Sievert and the team calculated that in one hour, a person in the middle of their simulated room receives a dose of about 56.1 nano-Sieverts, or .0000000561 Sieverts. That’s very very small. It takes about 1 whole Sievert to increase your chances of getting cancer by 1 percent.
The best way to get some perspective on what that means is to convert it to my new favorite unofficial unit of measurement, the Banana Equivalent Dose. As mentioned earlier, one of potassium’s isotopes, potassium-40, emits beta radiation and bananas are chock full of potassium, so a tiny portion of every banana is very slightly radioactive as a result. On occasion, large shipments of bananas have been known to set off radiation detectors at ports. When a person eats an average 150 gram banana, they absorb about .1 micro-Sieverts of radiation. A person would have to eat 27 bananas a day for 100 years to up their cancer risk by 1 percent. After developing their model, the team checked it by finding rooms in an apartment building that were similar in size and composition to what they simulated. The measured rates ended up being slightly higher than expected, about 67.7 nano-Sieverts per hour on average, but still largely in line with predictions. The researchers have submitted their work to the journals Chinese Physics C, and released a draft of it on the ArXiv.”