
“Annual values of Δ14C (in ‰) around the event”
LARGEST SOLAR STORMS in HISTORY (cont.)
https://linkinghub.elsevier.com/retrieve/pii/S0012821X25001827
https://phys.org/news/2025-05-reveals-strongest-solar-event-bc.html
New research reveals the strongest solar event ever detected, in 12350 BC
by University of Oulu / May 15, 2025
“An international team of scientists has discovered an extreme spike in radiocarbon corresponding to the year 12350 BC during the dusk of the last Ice Age. However, the strength of the event could not be assessed earlier because of the lack of an appropriate model. It has now been identified as the most powerful solar particle storm known to date—a colossal space weather storm that struck Earth 14,300 years ago. This recent finding expands the timeline and intensity of known solar activity and sets a new upper boundary for such solar phenomena.
In the study, postdoctoral researcher Kseniia Golubenko and Professor Ilya Usoskin at the University of Oulu, Finland, used their newly developed chemistry–climate model called SOCOL:14C-Ex, specifically designed to reconstruct solar particle storms under ancient glacial climate conditions. The model confirmed that the detected solar event was approximately 18% stronger than the notorious AD 775 event—until now the strongest solar storm ever recorded in tree-ring archives.
The work is published in the journal Earth and Planetary Science Letters. “Compared to the largest event of the modern satellite era—the 2005 particle storm—the ancient 12350 BC event was over 500 times more intense, according to our estimates,” says Dr. Golubenko. Other large known solar particle storms have occurred around 994 AD, 663 BC, 5259 BC and 7176 BC, and a few other candidates are under investigation. The new model was also verified using wood samples recently found in the French Alps, dating back some 14,300 years.

“Extreme solar storms (stars) known from cosmogenic isotopes, including the one of 12350 BC (the red star in the upper-left corner) and others as listed in Cliver et al. (2022). The red-shaded curve represents the decadal sunspot numbers (right-hand-side ordinate) reconstructed for the past nine millennia (Wu et al., 2018).”
Solar particle storms are rare, but when they occur, they bombard Earth with an enormous amount of high-energy particles. In comparison, the famous Carrington event in 1859 was a different kind of event and not accompanied by a solar particle storm. “The ancient event in 12350 BC is the only known extreme solar particle event outside of the Holocene epoch, the past ~12,000 years of stable warm climate,” says Golubenko. “Our new model lifts the existing limitation to the Holocene and extends our ability to analyze radiocarbon data even for glacial climate conditions.”
In the study, Golubenko and Usoskin designed the SOCOL:14C-Ex model to assess the solar particle storm intensity under glacial conditions. The model was successfully validated using data in tree rings from the AD 775 event and applied to the late Ice Age conditions to study the 12350 BC event. With the model, the researchers assessed the strength, timing, and terrestrial effects of the most extreme solar particle event presently known. The model—now validated under both Holocene and glacial conditions—marks a major step forward in analyzing radiocarbon variations across different climatic and geomagnetic epochs. The international research team included scientists from France and Switzerland, and was led by Professor Edouard Bard from CEREGE, France.
Solar particle storms can greatly enhance the normal production of cosmogenic isotopes like radiocarbon (14C) in the atmosphere by galactic cosmic rays. Such enhanced production, preserved in annual tree rings, serves as a clear cosmic timestamp making possible absolute dating of tree samples. Such dramatic spikes—known as Miyake events, named after the Japanese researcher who first discovered them—offer invaluable data for scientists studying both solar activity, ancient Earth systems and space climate.
“Miyake events allow us to pin down exact calendar years in floating archaeological chronologies,” says Usoskin. Radiocarbon signals from such events have already enabled researchers to precisely date Viking settlements in Newfoundland and Neolithic communities in Greece. The findings revise our understanding of solar physics and space weather extremes. “This event establishes a new worst-case scenario,” Golubenko notes. “Understanding its scale is critical for evaluating the risks posed by future solar storms to modern infrastructure like satellites, power grids, and communication systems.”
More information: Kseniia Golubenko et al, New SOCOL:14C-Ex model reveals that the Late-Glacial radiocarbon spike in 12350 BC was caused by the record-strong extreme solar storm, Earth and Planetary Science Letters (2025). DOI: 10.1016/j.epsl.2025.119383

“A contemporary magazine illustration depicted the radiant aura of 1859”
MIYAKE EVENTS
https://nature.com/articles/nature11123
https://science.org/marking-time-cosmic-ray-storms-can-pin-precise-dates-history
Past boon, future menace
by Michael Price / 13 April 2023
“Miyake events, blasts from the Sun or other stars that deposit spikes of radioactive carbon in tree rings, illuminate the past. But such a blast could seriously disrupt the present. The largest solar storm to hit our planet in recorded history was the Carrington Event from 1–2 September 1859, named after a British astronomer who linked a massive solar flare others had spotted with the bizarre phenomena that soon followed.
In the Northern Hemisphere, auroral displays were seen as far south as the Caribbean and were so bright in the northeastern United States that people could read newspapers at night. Telegraph stations caught fire as transmission wires sizzled under a barrage of electrons. Newspapers reported that birds began chirping in the night, and gold miners in California woke up in the wee hours to prepare breakfast, confusing the bright aurora for daybreak.
Yet compared with a Miyake event, those two scintillating days in 1859 are “barely a blip,” says Charlotte Pearson, a dendrochronologist and radiocarbon scientist at the University of Arizona. Carbon-14 records in tree rings from 1859 show virtually no upticks—nothing approaching the massive spike caused by Miyake events. A solar storm or gamma ray assault on the scale of the 774–75 C.E. event would similarly turn the night sky into an awe-provoking swirl of crackling, shimmering light.
It would also do catastrophic damage to technologies and infrastructure. “We’re massively vulnerable,” Pearson says. The surge of high-energy particles would fry satellite electronics, and astronauts in orbit likely would absorb lethal doses of radiation; even airplane passengers might absorb dangerous levels, Miyake says. As Earth’s magnetic field convulsed under the assault, surges of current would destroy power grids. Nearly anything running on electricity—from electric vehicles to cellphones to ventilators–would short out. At the very least, we’d experience widespread communications failures, she says.
The grave consequences have scientists trying to discern whether past Miyake events follow a pattern or cycle, or whether a future event would blindside us. “The goal is to try and understand the frequency and severity of these events with a view to actually do some kind of risk assessment,” says physicist Andrew Smith of the Australian Nuclear Science and Technology Organisation. That includes working out how to better shield people and equipment on Earth and in space. “Obviously, a big [Miyake] event today would be a disaster,” Smith says. “All of our tech would be in a smoking heap overnight.”

“Dr. Margot Kuitems (University of Groningen), seen here preparing samples
at the radiocarbon facility at the Centre of Isotope Research, Groningen”
MARKING TIME : TREE RINGS & ICE CORES
https://science.org/marking-time-cosmic-ray-storms-can-pin-precise-dates-history
Radiocarbon timestamps left in ancient tree rings by cosmic ray bombardments can date historical events with unprecedented precision
by Michael Price / April 13 2023
“One thousand years ago, according to Viking sagas, intrepid seafarers sailed west from Greenland to a coast they called Vinland. There they set up camps, harvested wild grapes, and skirmished with local people. At an archaeological site in a coastal town in Newfoundland called L’Anse aux Meadows, remnants of structures laid out like Viking longhouses and artifacts such as a bronze cloak pin and iron nails record their presence—likely the first Europeans to set foot in North America. Yet pinning down exactly when Vikings came to Vinland was impossible, until researchers discovered a kind of cosmic timestamp preceding their arrival.
In 2021, Margot Kuitems and Michael Dee of the University of Groningen obtained pieces of timber from a longhouse with visible tree rings and intact bark. Kuitems, an archaeologist, and Dee, a radiocarbon expert, were hoping to find a tree ring with an unusually large amount of carbon-14 (14C): the signature of a barrage of high-energy particles from outer space known to have occurred in 993–94 C.E. “It was a bit of a shot in the dark,” Kuitems says. But 28 rings in from the bark, they found the telltale 14C spike: persuasive evidence that Vikings had felled the fir and juniper trees used to build the longhouse in 1021 C.E. The timing of the Viking foothold at L’Anse aux Meadows heralds a revolution in archaeology: a newfound ability to whittle the age of wooden artifacts from decades down to single years. The breakthrough paving the way for such precision came in 2012, when Japanese physicist Fusa Miyake revealed that a massive influx of cosmic rays caused a big uptick in 14C in a tree ring dated to 774–75 C.E.
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/filer_public/89/44/894469bf-90c8-4b7a-a66b-7066bcb1bfbc/35-wood-lam-piece-studied-2_web.jpg)
“A piece of wood from the Norse deposit at L’Anse aux Meadows”
Since then, at least seven more confirmed spikes, known as Miyake events, have been found so far. The earliest well-supported spike dates to 7176 B.C.E. These chronological lighthouses are now guiding a growing cadre of scientists as they work to date ancient ruins, natural disasters, and other historic turning points. “If we’re able to start pinning things down to the year,” Dee says, “we can start analyzing early history, perhaps even prehistory, with the sort of rigor that previously we could only apply to modern history.” The technique “is phenomenally cool,” says Charlotte Pearson, a dendrochronologist and radiocarbon scientist at the University of Arizona’s Laboratory of Tree-Ring Research. “These are eureka moments, and we’re about to have a lot more of them.”
Even scribes in the early Middle Ages noticed the cosmic assault that produced Miyake’s first event. In 774 C.E., the Anglo-Saxon Chronicle, a collection of manuscripts that recounts key moments in Anglo-Saxon history, recorded the appearance in the sky, after sunset, of a “red crucifix.” Astronomers have speculated that the sighting may have been atmospheric dust scattering light from a nearby supernova, or an especially vivid aurora borealis triggered by a giant solar flare. That same year, thousands of kilometers to the east, as Japan’s Emperor Kōnin struggled with a succession crisis, a Japanese cedar on Yaku Island off the nation’s southern coast was sucking up carbon dioxide (CO2) through its short, bristled needles. The tree converted some of the CO2 into sugar and deposited that in the outermost layer of its wood. As empires rose and fell, the cedar added ring after ring of annual growth—succumbing only in 1956, at the venerable age of 1900 or so, to the blades of Yaku forestry managers.

A half-century later, Miyake, then a graduate student at Nagoya University, cut a cross-section of the cedar’s stump. Each of its rings held a trace of 14C. The radioactive isotope forms continuously in the upper atmosphere as cosmic rays—high-energy particles from space—collide with gas molecules, spawning neutrons. When one of these neutrons knocks out a proton in a nitrogen atom, that nitrogen is transformed into 14C. As it inhaled CO2, the cedar had incorporated the 14C into its wood. All green plants take up 14C and pass it on to the animals they sustain; it can be detected in fossils and other preserved tissues going back tens of thousands of years. Its mostly predictable decay rate is the basis of standard radiocarbon dating.

“Each tube in Fusa Miyake’s collection contains slices of
wood representing a single year of a tree’s growth.”
Using methods such as mass spectrometry, scientists can analyze organic remains to determine how much 14C has decayed since the life form that absorbed it died. That value is then compared against the 14C values of items with a known age—usually based on tree rings—giving the sample’s age to within a few decades under ideal circumstances. (Because the isotope has a half-life of about 5700 years, samples from earlier than about 50,000 years ago contain too little for dating.) But Miyake wasn’t trying to date the tree. She was looking for anomalous 14C spikes left by violent space weather events—solar flares, other outbursts from the Sun, and exploding stars—that unleash short, exceptionally intense showers of high energy particles.

“This chunk of wood (second image) came from the Japanese cedar in which
Miyake first found evidence of the 774-775 C.E. carbon-14 spike.”
Using techniques developed in part by Lukas Wacker, a physicist at ETH Zürich, she zeroed in on a period toward the end of the 8th century C.E., where previous radiocarbon scientists had detected an anomalous hump. Working ring by ring, she sliced out tiny wood chips from the cedar and ran them through an accelerator mass spectrometer to determine the ratio of 14C to stable carbon isotopes. In the ring corresponding to 774–75 C.E., she saw a 12% jump in 14C: an increase 20 times larger than ordinary cosmic ray oscillations produce. Other teams confirmed the spike in samples from a German oak and a New Zealand kauri tree.

“A decade ago, Fusa Miyake discovered two precise markers of time in the
rings of an ancient cedar: carbon-14 spikes now called Miyake events.”
Later that year, Miyake and her colleagues published a paper in Nature attributing the 14C spike to a massive bombardment of cosmic rays, perhaps from the Sun or a gamma ray burst from a distant star. In 2013, they found a second, slightly smaller 14C spike in the same Yaku cedar at 993–94 C.E. Other scientists started calling the phenomena Miyake events. When Wacker read about Miyake’s 774–75 C.E. and 993–94 C.E. spikes, he recognized the radiocarbon beacon’s untapped potential. He trained it on a chapel in Müstair, Switzerland, supposedly built by the first Holy Roman Emperor, Charlemagne, on a site where he and his party had survived a horrendous blizzard. Another team of scientists had dated the chapel to 785 C.E. by meticulously analyzing the widths of tree rings in a wooden beam—wider rings often indicate wetter years—then matching the patterns with more recent timber from the area until they had an unbroken record stretching from the present back to the beam.

“Pencil marks note specific years along tree rings from a Japanese cedar.”
Wacker knew that if his team could find the 14C spike from the 774–75 C.E. Miyake event in the beam, they could simply count to its outer edge to obtain a precise date. They did just that, confirming in 2014 the 785 C.E. date. “That was pioneering work,” Miyake says. “I was really happy to see that kind of application was made possible because of our findings.” While Wacker pursued the Holy Cross Chapel work, Dee and colleagues began exploring other ways that Miyake events might illuminate historical timelines confused or obscured by the passage of time.
Reliable written records—chronicles listing the exact years of empires and wars, and more mundane archives of taxes, trade receipts, births and deaths—go back only some 2500 years, Dee says, and only in societies that kept written records. Radiocarbon dates, with their decades or centuries of uncertainty, may help reconstruct history in broad strokes, but key historical moments often play out over shorter timescales. “One couldn’t examine the political events of the 20th century if you only had decadal resolution,” Dee says. “If the First World War and the Second World War had been in 1914 B.C. and 1939 B.C., we wouldn’t be able to tell those two things apart.”
In 2020, Kuitems and Dee used the technique to clear up a longstanding mystery surrounding rectangular ruins on a lake island in southern Siberia. Despite decades of archaeological work, nobody knew for sure how old the Por-Bazhyn site is, who built it, or what its purpose was. Por-Bazhyn lacked any sign of occupation such as trash heaps or charcoal from hearths, suggesting it was abandoned soon after construction. But the 774–75 C.E. spike was present in an outer ring of a beam, indicating the source tree had been felled in 777 C.E. That was during the reign of Tengri Bögü Qaghan, a Uyghur monarch who converted to Manichaeism, a religion that views existence as an epic struggle between good and evil.
Most likely, Kuitems says, Bögü Qaghan built Por-Bazhyn as a Manichaean monastery. Por-Bazhyn’s precise age points to the apparent reason it was never occupied. In 779 C.E., rebels opposed to Manichaeism killed Bögü Qaghan. “So at the moment this whole building was finished, it immediately became useless,” Kuitems says. Dee also contributed to work by German colleagues that used the same Miyake spike to date another ancient fortress, on an island in Lake Āraiši in central Latvia. “These were questions that people couldn’t answer for decades, and now we could with this method,” Kuitems says.

“A Miyake event in timber from Viking structures in Newfoundland gave
them a precise date of 1021 C.E. (These are modern reconstructions.)
In the wake of their headline-grabbing Vinland success, the Groningen team is tackling another alluring set of historical puzzles: “floating” chronologies, or ancient, internally consistent timelines that are untethered to specific dates. For instance, Dee has long hoped to moor the floating timelines of ancient Egypt’s pharaohs to our modern calendar. Radiocarbon dating has pegged the Egyptian chronology to within a couple hundred years; scientists have tried to use ancient observations of known astronomical events, such as star and planet alignments, to narrow dates further. But timelines remain blurry. Egypt’s Old Kingdom era, for example, encompasses dozens of pharaohs who reigned from approximately 2700 to 2200 B.C.E. “The calendrical dates are not even really known to the century,” Dee says.
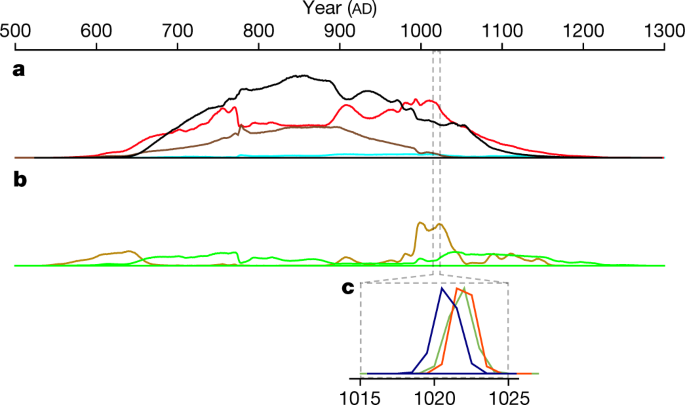
“a, b, Averaged probability density functions for different sample types, c, Wiggle-matched probability density functions for the last growth ring of each wood item.”
Many native Egyptian tree species don’t produce annual growth rings, complicating the quest for Miyake events there. But the Egyptians imported conifers—which builders often preferred for their straightness and strength—from Lebanon and Syria. The pandemic and laws restricting the export of Egyptian artifacts have slowed his team’s work, Dee says. But he has his sights set on discovering a precise date for the construction of the Great Pyramid, which was built by the Old Kingdom Pharaoh Khufu. “There is an old Arab proverb which goes, ‘Man fears time; time fears the Pyramids,’” Dee says. “If this structure, and his reign, could be precisely dated, that would solve a mystery that has endured since antiquity itself.”
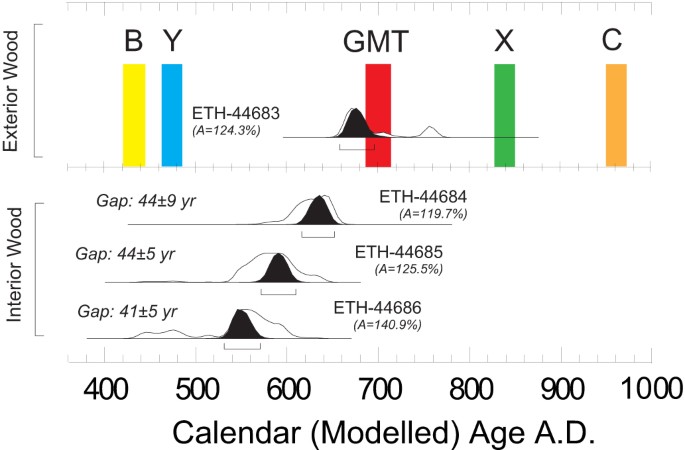
“Posterior probability distributions of 14C dates from Beam e, Lintel 3 (Temple I)”
Another floating chronology Dee’s team hopes to anchor is the Mesoamerican Long Count timekeeping system. Used by the Maya, Aztec, and other Mesoamerican civilizations, the system counts the days linearly from a supposed day of the world’s creation. While there have been several attempts to correlate the Long Count with the Gregorian calendar, academics are still debating the precise creation date. Finding a Miyake event in wood from a Mesoamerican structure—such as a lintel in the Maya temple Tikal in Guatemala, whose construction is recorded in the Long Count—would settle the matter. Miyake events also promise to date natural disasters that have altered the course of human history. “This technique can help answer questions about the rise and fall of civilizations,” Pearson says.
In 2017, Clive Oppenheimer, a volcanologist at University of Cambridge, teamed up with Wacker and others to use a Miyake event to date a larch tree buried in ash by the so-called Millennium eruption of Mount Paektu, a volcano straddling the border of China and North Korea. Radiocarbon dates pegged the eruption—one of the biggest in the past 10,000 years—to sometime during the ninth to 11th centuries. Some historians hypothesized that the blast led to the fall in 926 C.E. of the Bohai Kingdom, which encompassed parts of the Korean Peninsula, Northeast China, and Russia’s Far East. Oppenheimer and colleagues used the 774–75 C.E. Miyake event to date the larch’s demise to 946 C.E., exonerating the eruption in the kingdom’s collapse. “Thanks to the Miyake event, we could really nail it,” Oppenheimer says. “With anything that kills trees—earthquakes, fires, biological pests—there’s now the potential to date that very, very precisely.”
Pearson is hoping to replicate a version of that success in a tree that was downwind of the volcano Thera when it erupted around 3500 years ago on the island of Santorini, possibly contributing to the fall of the Minoan civilization. The widespread devastation would have disrupted commerce throughout the Mediterranean and sent refugees scrambling for new homes. A 14C spike centered on 1528 B.C.E.—so far unconfirmed as a Miyake event—confirms that in 1562 B.C.E, plus or minus 1 year, an abrupt chemical signal shows up in the tree’s rings, possibly caused by sulfate from the eruption. Miyake events may even shed light on potential future cataclysms. Dendrochronologist Bryan Black, also at the Arizona treering lab, recently used the 774–75 C.E. Miyake event to date two mass tree deaths separated by more than 80 kilometers in the Puget Sound region of the U.S. Pacific Northwest.

“A shows annual 14C ages (radiocarbon years B.P.) from two calendar-dated
tree-ring series. B shows the annual 14C ages in A converted to Δ14C (21)
to emphasize patterns used to synchronize the floating chronology.”
The forests grew along two distinct shallow faults near present-day Seattle, and geological evidence shows they died more than 1000 years ago. Sediments record the upheaval, as do oral traditions of the indigenous Salish people. Earth scientists have wondered whether both faults ruptured separately, or at once in a single massive earthquake. Black found these trees all died during the same season between 923 and 924 C.E., suggesting the faults could conspire again to produce a massive earthquake. “It takes the current worst-case scenario of these shallow faults and raises it to the next level,” he asserts.
Radiocarbon scientsists are scouring tree ring archives for more Miyake events. Kuitems hopes to firm up one in the sixth millennium B.C.E., which may allow her to precisely date an undisclosed Neolithic site in Europe. Other researchers have spotted a possible Miyake cluster in 1261 C.E., 1268 C.E., and 1279 C.E. Several different labs are aiming to verify these and other 14C spikes in trees from around the world. “It’s like a sort of jigsaw puzzle,” Dee says. “In the end, if we have enough spikes, we’ll be able to put together a lot of this history of the Holocene in absolute time.”
Ice core data can also confirm Miyake events. Cosmic rays spawn two other radioisotopes—beryllium-10 and chlorine-36—in the upper atmosphere. Rather than lodging in living tissue, these isotopes are washed to the surface in rain and snow, and they can end up in the annual layers of polar ice sheets. Like 14C in tree rings, sharp spikes of beryllium-10 and chlorine-36 in the ice have been correlated to known Miyake events. At a radiocarbon conference in Zürich last year, physicists Andrew Smith and David Fink of the Australian Nuclear Science and Technology Organisation reported isotopic spikes at 774–75 C.E. and 993–94 C.E. in Antarctic ice cores. Continued analysis of the cores could reveal other, previously unknown Miyake events that dendrochronologists could follow up on.
Miyake, too, is continuing her quest to find more of the events that bear her name. She has joined forces with Arizona’s Laboratory of Tree Ring Research, whose 700,000 tree ring samples–the largest collection in the world–offer an unparalleled record of past climate and cosmic events. There, Miyake and her Arizona colleagues, along with colleagues around the world, are aiming to patch together from recently deceased and long-dead trees a complete radiocarbon record stretching back 12,000 years. Filling in the gaps between Miyake events is also important, says Pearson, as they contain solar patterns that can be matched with tree-ring and ice-core records to improve radiocarbon dating. Pearson and Wacker are preparing to submit another 1100 years’ worth of data from California’s bristlecone pines, which can live for more than 4000 years, to an international radiocarbon calibration repository. Miyake is looking to samples of other long-lived trees from Russia, Finland, and Japan, and has already spotted several as-yet unpublished Miyake events, she says. Miyake’s pioneering analysis of the cedar continues to ripple through studies of the past, colleagues say. “She trusted her instincts and was so confident in what she had that she was able to convince us all that there was a real effect,” Dee says. “And the rest, as they say, is history.”
PREVIOUSLY
a CARRINGTON EVENT
https://spectrevision.net/2010/05/12/a-carrington-event/
CHARGED PARTICLES
https://spectrevision.net/2018/05/30/electric-dark-matter/
LIGHTNING SCARS
https://spectrevision.net/2018/10/31/lightning-scars/
SPACE WEATHER and REVOLUTIONS
https://spectrevision.net/2018/12/10/space-weather-and-revolutions/
IONOSPHERE MAPPING
https://spectrevision.net/2024/12/19/ionosphere-mapping/
SOLAR SUPERFLARES
https://spectrevision.net/2023/05/10/solar-superflares/



